Herpes Labialis Curtailed by 100 Year Old Vaccine
While the world awaits the approval of a herpes vaccine, a very old vaccine was recently found to offer some protection against this sexually transmitted disease (STD).
As of December 1, 2023, various studies have found the bacillus Calmette-Guérin (BCG) vaccine offers beneficial off-target effects that might impact the recurrence of herpes simplex virus (HSV) infections in certain people.
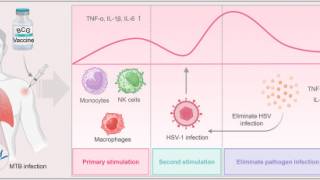
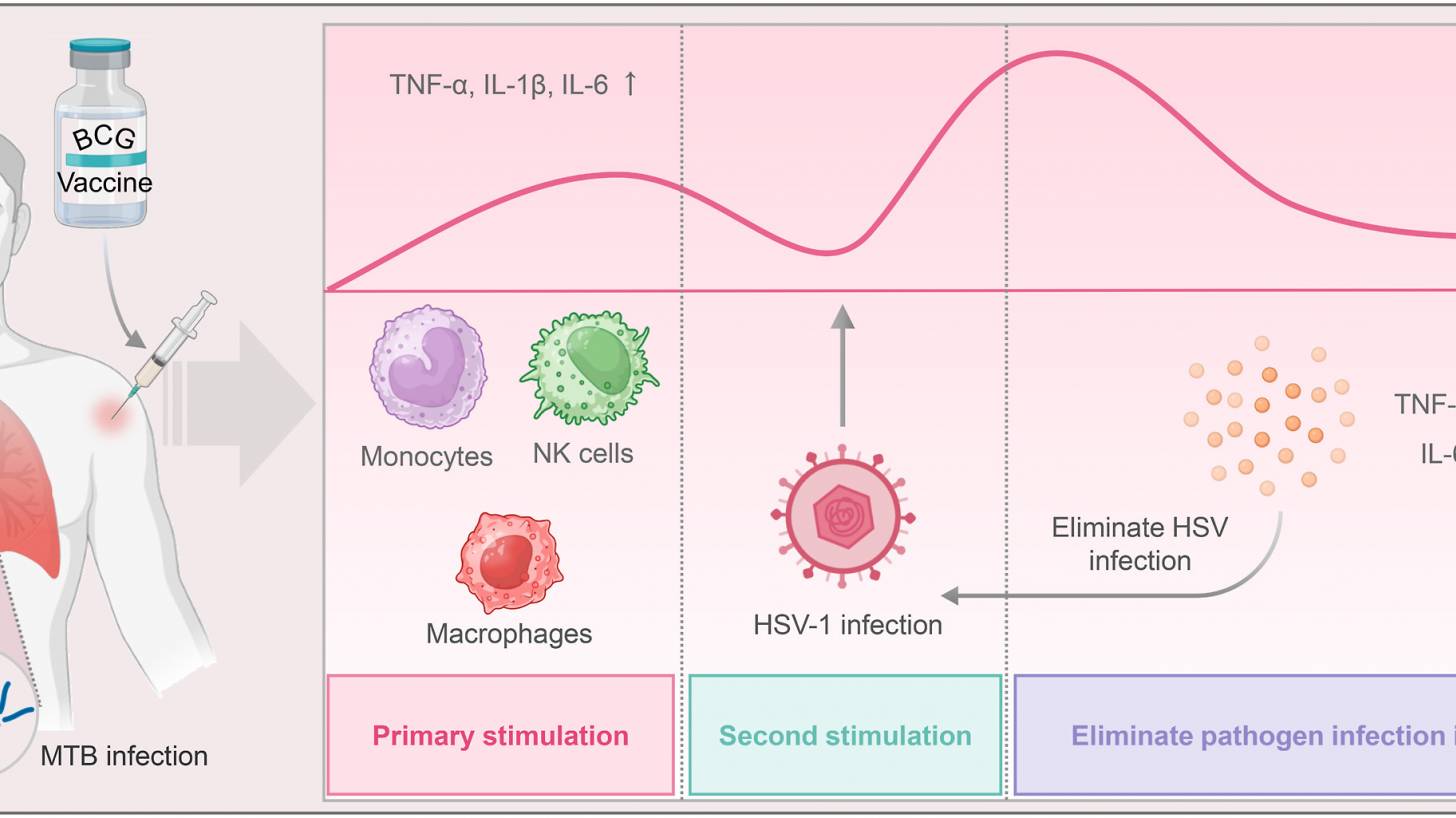
Previous research suggests that BCG vaccination induces three non-specific immune responses: trained immunity, heterologous immunity, and anti-inflammatory effects. These immune responses have been used to prevent and treat various diseases, including HSV-1.
A Commentary published by eClinicalMedicine in October 2023 summarized a phase 3 clinical trial conducted across multiple countries to assess the impact of BCG vaccination on recurrent herpes labialis, known as cold sores.
Excerpts from this article are inserted below:
In this nested randomized controlled clinical trial within the multicentre, phase 3 BRACE study, 6,828 healthcare workers were randomized in 36 sites in Australia, the Netherlands, Spain, the United Kingdom, and Brazil to receive BCG-Denmark or no BCG vaccinations.
The results demonstrated that, over a 1-year follow-up period, the BCG-Denmark vaccinated group experienced reduced duration, frequency, severity, and impact on quality of life related to recurrent herpes labialis than the control group.
Furthermore, in the BCG-immunised individuals, the time to the first recurrence was extended by 1.55 months compared with the control group (p = 0.02), indicating the potential benefits of BCG vaccination for individuals with herpes labialis (p = 0.003).
However, a subgroup analysis revealed that the benefits of BCG vaccination were primarily observed in male participants.
Previously, a systematic review published in 2020 also demonstrated the benefits of BCG vaccination for 78% of adult patients with recurrent genital or herpes labialis, with 37% experiencing long-term remission and a reduction in outbreak frequency or severity by 41%.
These researchers encouraged a cautious interpretation of the clinical trial results, which is necessary, and the study authors acknowledge these limitations in their manuscript.
Firstly, the study had a relatively small sample size, with only 38 individuals receiving BCG vaccination. Secondly, the participants' immune status during the study was not considered.
Nonetheless, the study by Pittet and colleagues is a unique and innovative contribution to the literature. It compared the protective effect of BCG vaccination against recurrent herpes labialis between sexes and suggested an increased risk of primary cold sore occurrence following BCG vaccination.
These results imply that BCG-induced non-specific immune responses can enhance the body's immune response but may also exacerbate existing inflammatory or immune-related diseases.
The Bill & Melinda Gates Foundation and others funded this study.
In the United States, access to Merck's TICE® BCG vaccine is limited and is targeted against tuberculosis and TB meningitis.
Moreover, herpes vaccine candidates continue conducting clinical research in December 2023.
Our Trust Standards: Medical Advisory Committee