Pneumococcal Vaccines May Reduce Antibiotic and Ear Tube Use

A small observational study has confirmed that the introduction of Pneumococcal Conjugated Vaccines (PCV) has been successful in reducing the number of invasive and lower respiratory pneumococcal infections in children.
The impact of the vaccines on upper respiratory infections caused by pneumococci was reported to be less clear.
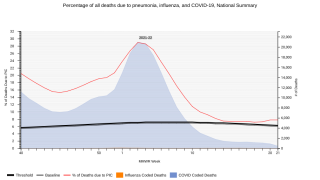
Streptococcus pneumoniae, the pneumococcus, is a respiratory bacterial pathogen, well known for its association with pneumonia and meningitis.
In this Danish population-based retrospective observational study, these researchers used proxies for respiratory infections in children, such as antibiotic use and ventilation tube insertions (VTI), to estimate the impact of the vaccine on a national level.
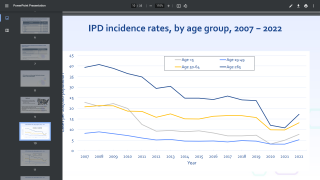
The study was designed to compare trends in the incidence rate of antibiotic prescriptions and VTIs in the period 2000–2014, where PCV7 was introduced in 2007 and PCV13 in 2010.
The introduction of PCV7 and PCV13 correlated with changes in the incidence rate from an almost steady increase in prescription of antibiotics in the pre-PCV period, to a decreasing incidence for all children age 0–15 years.
This data indicates that the integration of pneumococcal vaccines in the Childhood Vaccination Programme likely reduced the incidence of upper respiratory diseases due to pneumococci in Denmark.
Although pneumococcal vaccination coverage is increasing globally, it is nowhere near optimal vaccination rates.
The World Health Organization (WHO) estimates that globally S. pneumoniae kills close to 500,000 children less than 5 years old annually, with most of the deaths occurring in developing countries.
The CDC recommends vaccination with the pneumococcal conjugate vaccine for all babies and children younger than 2 years old, all adults 65 years or older, and people 2 years through 64 years old who are at increased risk for pneumococcal disease due to certain medical conditions.
According to the CDC, there is no evidence on the safety of PCV13 use during pregnancy. As a precaution, women who need the vaccine should be vaccinated before becoming pregnant, if possible.
There are two types of pneumococcal vaccines, conjugate vaccines and polysaccharide vaccines. They are given by injection either into a muscle or just under the skin.
The CDC recommends vaccination with the pneumococcal conjugate vaccine PCV13 or Prevnar13 for:
- All babies and children younger than 2 years old
- All adults 65 years or older
- People 2 through 64 years old who are at increased risk for disease due to certain medical conditions
Separately, the CDC recommends vaccination with the pneumococcal polysaccharide vaccine PPSV23 or Pneumovax23 for:
- All adults 65 years or older
- People 2 through 64 years old who are at increased risk for disease due to certain medical conditions
- Adults 19 through 64 years old who smoke cigarettes
The CDC Vaccine Price List provides private sector vaccine prices for general information.
Vaccine discount information can be found here.
These researchers did not disclose conflicts of interest: Michael Frantz, Howitza Zitta, Barrella HarboebcHeleneIngelsd, Palle Valentiner-Branthe, Kåre MølbakeBjarki, Ditlev Djurhuusfg.
Our Trust Standards: Medical Advisory Committee
- A nationwide study on the impact of pneumococcal conjugate vaccination on antibiotic use and ventilation tube insertion in Denma
- Impact of pneumococcal conjugate vaccine (PCV7 and PCV13) on pneumococcal invasive diseases in Italian children and insight into
- Safety, reactogenicity and immunogenicity of two investigational pneumococcal protein-based vaccines: Results from a randomized
- Pneumococcal Vaccination: What Everyone Should Know
- Pneumococcal Vaccine Recommendations
- Licensure of a 13-Valent Pneumococcal Conjugate Vaccine (PCV13) and Recommendations for Use Among Children --- Advisory Committe
- Pneumococcal Conjugate Vaccines May Reduce Children Ear Infections