Vaccines Extraordinarily Reduce Pneumococcus Impact

At the beginning of the 20th Century, South African miners often were in poor health, and infections such as influenza and pneumonia quickly spread, killing up to one in ten miners yearly.
The biggest killer was pneumococcal pneumonia, wrote GAVI on May 16, 2023.
Within the first four months of working in the mines, 1–2% of men had been infected with it, and among men proven to be infected, 25–56% later died.
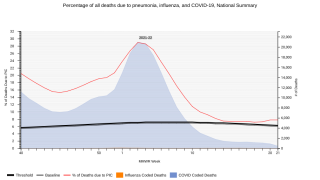
They weren't alone. A century later, pneumococcus bacteria continues to be the leading cause of severe pneumonia and bacterial meningitis worldwide.
Pneumonia kills some 740,000 children under five each year, making it the single most significant infectious cause of death in children.
Case fatality rates for children with severe and invasive pneumococcal infections, such as bacteremia or meningitis, range from 20–50% in low- and middle-income countries.
However, pneumococcal pneumonia is deadly no matter who is affected or where a person lives.
Introducing pneumococcal conjugate vaccines (PCVs) has substantially reduced the burden of death and disease, but millions of children remain unvaccinated.
And existing vaccines don't protect against all 90 pneumococcus strains.
Efforts to create a vaccine against pneumococcus began in 1911 when South African mining representatives approached a British doctor called Almroth Wright.
Wright adopted an approach against pneumococcus: injecting whole heat-killed bacteria into thousands of migrant laborers working in gold mines near Johannesburg.
The results of these initial vaccine trials were encouraging – suggesting a substantial reduction in pneumonia cases and deaths after vaccination.
However, it soon became apparent that pneumococcus wasn't a single enemy but that there were multiple 'serotypes' – groups within a species of microorganism that share distinctive surface structures.
Once researchers had figured out how to identify them, they combined these whole killed bacteria into multivalent vaccines to protect against more than one pneumococcus serotype.
During the 1930s and '40s, researchers began to test polysaccharide-based pneumococcal vaccines (PPVs) derived from the polysaccharide "capsule" that pneumococcus wears as a kind of coat. This capsule varies between serotypes, and researchers discovered it was a vital part of the bacterium that people's immune systems responded to following inoculation.
However, with the discovery of penicillin, interest in pneumococcal vaccines waned, and their development stalled.
In 1977, Merck licensed a PPV vaccine containing capsular polysaccharides from 14 pneumococcus strains, followed in 1983 by one containing 23 capsular polysaccharides – Pneumovax – still in use today.
According to a meta-analysis published in 2008, which combined the results of multiple trials, PPVs reduced the incidence of all invasive pneumococcal disease in adults worldwide by 82%.
However, PPV vaccines are largely ineffective in children younger than two years old, presumably due to their immature immune systems.
To overcome this problem, scientists needed to find a way to boost the immune response.
So, they began chemically attaching these capsular polysaccharides to an inactivated form of the diphtheria toxin, called a toxoid, which the immune system easily recognizes.
A pivotal moment came in 2000, with the publication of a clinical trial that evaluated the first of these pneumococcal conjugate vaccines (PCVs) – which was designed to protect against the seven most common pneumococcal strains – in 37,868 infants in Northern California.
It found that the incidence of pneumococcal meningitis was reduced by more than 95% in fully vaccinated infants.
And the efficacy against all invasive pneumococcal disease was greater than 97%.
This vaccine, known as PCV7 or Prevnar7, was licensed in 2000.
However, pneumococcus was not so easily defeated.
Effective as PCV7 was against the strains it covered, its routine use began to be associated with an increased incidence of disease caused by a different strain, called serotype 19A – a phenomenon known as "serotype replacement."
This highlighted the value of vaccinating against a broader range of strains.
PCVs covering 10, 13, and 15 strains have since followed; in some cases, the polysaccharides are conjugated to diphtheria toxoid; in others, to tetanus toxoid or protein D.
In 2021, PCVs protecting against 15 and 20 pneumococcal strains were approved for adult use in the U.S., followed by European authorization in late 2021/early 2022.
PCVs covering more than 20 strains are also in development.
Three PCVs are currently prequalified by the World Health Organization (WHO) for use in infants and children: PCV13, manufactured by Pfizer; and two PCV10 vaccines, manufactured by GSK and the Serum Institute of India.
WHO recommends the inclusion of PCVs in childhood immunization programs worldwide, and as of the end of 2020, 148 out of 194 WHO member states had introduced it – including 63 Gavi-eligible lower-income countries.
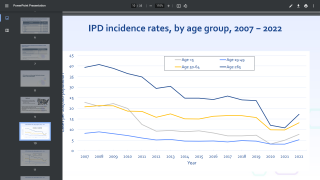
These childhood vaccination programs have had an extraordinary impact on rates of pneumococcal disease in those countries that have introduced them.
Routine childhood immunization against pneumococcus has also reduced the burden of pneumococcal disease in older children and adults who haven't been vaccinated against it – presumably because there's less community transmission of strains covered by the vaccines.
Four vaccines are currently available for preventing adult pneumococcal disease: PCV13, PCV15, PCV20, and the polysaccharide-based pneumococcal vaccine, PPV23.
If PCV13 or 15 are used, this is usually followed by a dose of PPV23, typically a year later.
Adults who receive PV23 or PCV20 usually only need one dose.
The U.S. CDC updated its recommendation in February 2023.
The road to defeating pneumococcus has been a long one, and there's still a way to go, but through sustained efforts to increase vaccine uptake and future innovations, many more lives could be saved, wrote GAVI.
The unedited GAVI article is posted at this link.
Precision Vaccinations publishes pneumococcal vaccine news.
Our Trust Standards: Medical Advisory Committee