$3 Million Vaccine Development Awarded To Combat Pneumococcus

A St. Louis bioscience startup announced it has received a $3 million federal grant to advance the company’s development of a next-generation vaccine to combat pneumococcus.
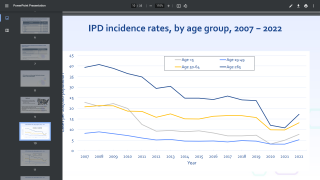
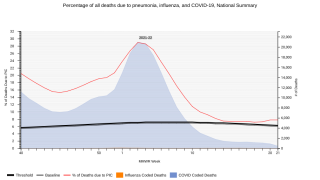
Pneumococcus is one of the most common causes of severe pneumonia.
In addition, pneumococcus can cause other types of infections, such as bloodstream infections, pneumonia, meningitis, and middle ear infections, says the Centers for Disease Control and Prevention (CDC).
VaxNewMo said in a press release on February 21, 2020, it has earned a Phase II Small Business Innovation Research (SBIR) grant, which is awarded to companies to conduct research and development that can more quickly lead to private sector commercialization.
And, this award further shows the commitment and support of the National Institutes of Health for VaxNewMo’s bioconjugation technology for vaccine development.
“This coveted SBIR grant will allow VaxNewMo to advance the development of a next-generation, multivalent pneumococcal bioconjugate vaccine that can cover the world’s most frequently-encountered pneumococcal serotypes at a price tag that is affordable for all countries,” said Christian Harding, Ph.D., VaxNewMo CEO, in the press release.
“We will use the Phase II SBIR grant to establish processing capabilities to manufacture the vaccines.”
Previously, the company published a research article, September 2019 that discussed ‘bioconjugate vaccines targeting the capsule of hvKp that can provide immunity and protection against extremely lethal hvKp strains. Additionally, they discussed that bioconjugation is a promising technology for the rapid development of efficacious vaccines against emerging bacterial threats.’
The new funding also will be used to hire three new scientists for VaxNewMo’s lab inside the BioGenerator Labs.
In the USA, there are 2 kinds of pneumococcal vaccines available today. These vaccines protect against many, but not all types of pneumococcal bacteria.
An unrelated study published in Nature during February 2019, said ‘currently licensed pneumococcal conjugate vaccines are synthesized chemically, which is a laborious process plagued with technical challenges, low yields, and batch-to-batch variations, highlighting the need for improved conjugate vaccine synthetic methodologies.’
This means there is still a chance someone can develop the pneumococcal disease after vaccination, says the CDC.
VaxNewMo is an early stage biotechnological company developing next-generation conjugate vaccines against infectious diseases using our proprietary in vivo conjugating technology. Our platform technology supports the generation of polysaccharide-protein conjugate based vaccines against multiple targets.
Vaccine Development news published by Precision Vaccinations.
Our Trust Standards: Medical Advisory Committee