Pfizer Completes Third Phase Three 20vPnC Vaccine Study

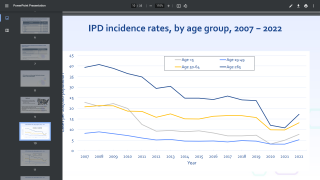
A 20-valent pneumococcal conjugate vaccine candidate was found to elicit responses across 20 serotypes, meeting the primary immunogenicity objective of an extensive clinical trial.
In this phase 3 study, the 20-valent pneumococcal polysaccharide conjugate vaccine (20vPnC) safety profile was reported similar to the Prevnar 13® control group.
This clinical lot consistency study is expected to satisfy licensure requirements for manufacturing consistency by the U.S. Food and Drug Administration (FDA), and other countries’ regulatory agencies.
New York based Pfizer Inc. announced top-line study results on May 14, 2020, which described the safety and evaluated the consistency of immune responses elicited across three different lots of its 20vPnC vaccine candidate, in adults 18 through 49 years of age, not previously vaccinated against pneumococcal disease.
“We are excited by the progress of our adult development program for 20vPnC as this is the second phase 3 trial for this investigational vaccine for which we have positive topline data,” said Kathrin U. Jansen, Ph.D., Senior Vice President and Head of Vaccine Research & Development, Pfizer, in a press statement.
“These new data highlight Pfizer’s strong heritage, expertise, and success in manufacturing highly-complex biological products such as pneumococcal conjugate vaccines. Demonstration of lot consistency is critical to help ensure that vaccine recipients receive the same level of protection irrespective of the manufactured lot used.”
Pfizer will seek to present and publish outcomes from this clinical trial at a future date once safety and immunogenicity data have been fully analyzed.
Pfizer’s 20vPnC vaccine candidate includes 13 serotypes already included in Prevnar 13.
Pfizer’s Phase 3 adult clinical program for 20vPnC includes 3 clinical trials (NCT03760146, NCT03828617, and NCT03835975) evaluating the vaccine candidate for the prevention of invasive disease and pneumococcal pneumonia in adults 18 years or older.
Combined, these trials have enrolled more than 6,000 adult subjects, including populations of vaccine-naïve adults and adults with prior pneumococcal vaccination. All three trials have been completed.
This new press release refers to NCT03828617: Phase 3 randomized, double-blind trial enrolled 1,700 adults aged 18 through 49 years with no history of pneumococcal vaccination.
Additional trials include NCT03760146: Phase 3 randomized, double-blind trial comparing immune responses in patients ≥60 years old after 20vPnC administration to responses in a control group receiving 13vPnC or PPSV23.
And, NCT03835975: Phase 3 randomized, open-label trial, designed to describe the safety and immune response of 20vPnC in an estimated 875 adults aged 65 years or older with prior pneumococcal vaccination.
The 7 new serotypes included in 20vPnC are global causes of invasive pneumococcal disease and are associated with high case-fatality rates, antibiotic resistance, and/or meningitis.
Together, the 20 serotypes included in 20vPnC are responsible for the majority of currently circulating pneumococcal disease in the U.S. and globally, says Pfizer.
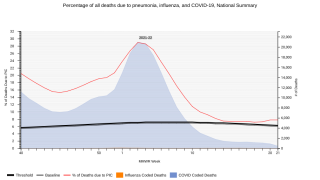
Pneumococcal disease is a name for any infection caused by bacteria called Streptococcus pneumoniae, or pneumococcus, says the US Centers for Disease Control and Prevention (CDC).
Pneumococcal infections can range from ear and sinus infections to pneumonia and bloodstream infections. There are vaccines to help prevent pneumococcal disease.
Globally, pneumococcal pneumonia is estimated to cause about 500,000 deaths and 30 million episodes in adults 70 years and older annually.
Pneumococcal disease is common in young children, but older adults are at the greatest risk of serious illness and death. Vaccines are the best way to prevent pneumococcal disease.
There are 2 vaccines used in the USA helping to protect people against pneumococcal disease:
- Pneumococcal conjugate vaccine (PCV13)
- Pneumococcal polysaccharide vaccine (PPSV23)
Most people who get a pneumococcal vaccine do not have any serious problems with it. However, side effects can occur. Most side effects are mild, meaning they do not affect daily activities, says the CDC.
Pneumococcal disease vaccine development news published by Precision Vaccinations.
Our Trust Standards: Medical Advisory Committee