Clearing Up Pneumococcal Vaccine Questions

A new commentary published in JAMA reinforces the changes by the Centers for Disease and Control (CDC)’s vaccine committee in 2019 regarding pneumococcal vaccine recommendations for seniors.
This news article was written by Bridget M. Kuehn, MSJ, on January 14, 2020, saying the CDC’s Advisory Committee on Immunization Practices (ACIP) suggests that adults aged 65 years or older should receive only the 23-valent pneumococcal polysaccharide vaccine (PPSV23) with a few exceptions.
This ACIP announcement was issued on November 22, 2019, because the pneumococcal vaccination recommendations had undergone several changes, causing confusion for those who need to select a preventive vaccine.
In 2014, the ACIP recommended that older adults receive the 13-valent pneumococcal conjugate vaccine (PCV13) first, then the PPSV23 at least 1 year later.
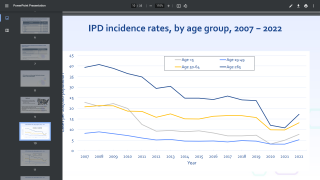
Since then, widespread childhood vaccination with PCV13 and its predecessor the 7-valent pneumococcal conjugate vaccine have “led to sharp declines in pneumococcal disease among unvaccinated children and adults,” according to the ACIP’s review of the evidence.
Based on that review, the ACIP voted to remove the recommendation for routine use of PCV13, followed by PPSV23 in this senior population.
“Incidence of PCV13-type disease has been reduced to historically low levels among adults aged ≥65 years through indirect effects from pediatric PCV13 use,” the authors wrote.
The PPSV23 vaccine contains 12 serotypes in common with PCV13 and an additional 11 serotypes, for which there are no indirect effects from PCV13 use in children.
But, adults aged 19 years or older with an immunocompromising condition, cerebral spinal fluid leak, or cochlear implant should continue to receive both vaccines, the ACIP recommends.
And, some adults aged 65 years or older may have a greater risk of exposure to strains of Streptococcus pneumoniae covered by the PCV13 vaccine, such as those seniors living in nursing homes or long-term care facilities, people living in areas with low rates of pediatric vaccination with PCV13, or those traveling to areas with no childhood PCV13 vaccination program.
Furthermore, the ACIP recommends that healthcare providers engage in shared decision-making with such at-risk patients to decide whether they should continue to receive PCV13 followed by the PPSV23 vaccination.
“ACIP will continue to review relevant data as they become available and update pneumococcal vaccination policy as appropriate,” the CDC’s authors wrote.
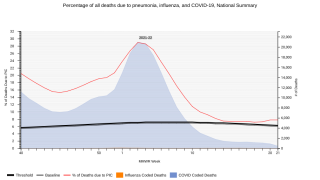
This ACIP recommendation is important since about 175,000 cases of the pneumococcal disease occur each year in the USA. And, the fatality rate is between 5-7 percent, says the CDC.
The pneumococcal disease is spread from person to person by droplets in the air. The pneumococci bacteria are common inhabitants of the human respiratory tract.
There are 2 major clinical syndromes of invasive pneumococcal disease: bacteremia, and meningitis. They are both caused by infection with the same bacteria but have different manifestations.
Pneumococcal pneumonia is the most common disease caused by pneumococcal infection and can occur in combination with bacteremia and/or meningitis, or it can occur alone.
Moreover, there are 3,000-6,000 cases of pneumococcal meningitis each year.
Additionally, new research is helping to clarify the benefits of childhood vaccinations.
A study published on October 15, 2019, found a bacterial infection that can lead to pneumonia or meningitis is also linked to weakened immune systems in children. This study found the rate of immune deficiency in healthy children presenting with the pneumococcal disease was up to 26.4 percent.
To address this issue, the CDC continues to say all infants should be given a primary series of PCV13, at ages 2, 4, and 6 months with a booster at age 12 to 15 months.
The CDC’s recommendation of the PCV13 vaccine, (Prevnar 13) and pneumococcal polysaccharide vaccine (PPSV23, Pneumovax) should not be given at the same visit.
Since these vaccination suggestions have changed and could be confusing, the CDC suggests everyone speak with a healthcare provider to learn which vaccine is best for their needs.
Pneumococcal disease and vaccine news published by Precision Vaccinations.
Our Trust Standards: Medical Advisory Committee