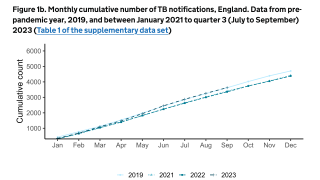
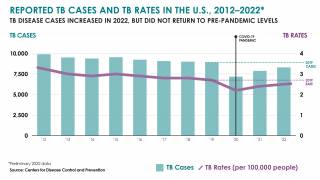
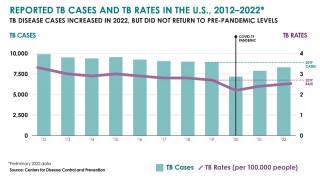
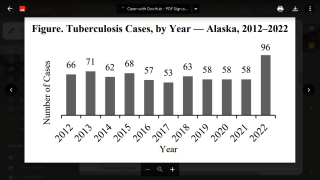
$30 Million Dollars to Accelerate Tuberculosis Vaccine Development

A team of collaborators received the first installment totaling $30 million dollars in its 1st year from the National Institute of Allergy and Infectious Diseases of the National Institutes of Health (NIH), to establish 3 immunology research centers to accelerate Tuberculosis (TB) vaccine development.
As the team reaches its milestones, the contract will amount to $60 million dollars over 7-years.
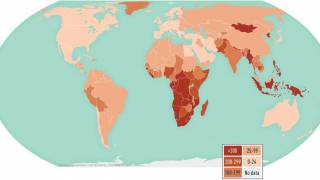
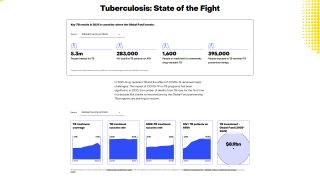
This effort is important since tuberculosis is a leading cause of death from an infectious disease in the world. Nearly 1.7 billion people globally are infected with Mycobacterium tuberculosis (Mtb), a germ spread by airborne transmission.
In 2017, about 1.6 million died from TB, according to the NIH.
W. Henry Boom, M.D., director of the TB Research Unit and professor of medicine at the Case Western Reserve University School of Medicine and chief of the Division of Infectious Diseases & HIV Medicine at University Hospitals Cleveland Medical Center, is a co-principal investigator (PI) for the program.
Additional co-PIs for the program are Sarah Fortune, M.D., professor at the Harvard T.H. Chan School of Public Health, and JoAnne Flynn, Ph.D., professor at the University of Pittsburgh.
These research centers are also called "Immune Mechanisms of Protection against Mycobacterium tuberculosis (IMPAc-TB) Centers." This program aims to make an impact on a global scale by accelerating TB vaccine development.
Researchers will study immune responses needed to prevent initial infection, establish latent infection, and transition to active TB. Every aspect of research from basic, to translational, to clinical science will be involved in this project.
"Our purpose is to breakdown the silos that exist between basic science and translational sciences and animal models and human disease," Dr. Boom said.
"My collaborators will use cutting-edge molecular and cell biology as well as animal models, and my role will be to build on that research at a global, human scale with clinical studies in London, Uganda, and South Africa."
Because TB isn't prevalent in the United States, human research must have a global footprint, Dr. Boom said.
"As we discover what's driving this epidemic and work to reach underserved populations that have poor public health."
This project has been funded by the National Institute of Allergy and Infectious Diseases, National Institutes of Health, Department of Health and Human Services, under Contract 75N93019C00071.
Case Western Reserve University is one of the country's leading private research institutions.
Tuberculosis (TB) information published by Precision Vaccinations
Our Trust Standards: Medical Advisory Committee