$550 Million Supports New TB Vaccine Development
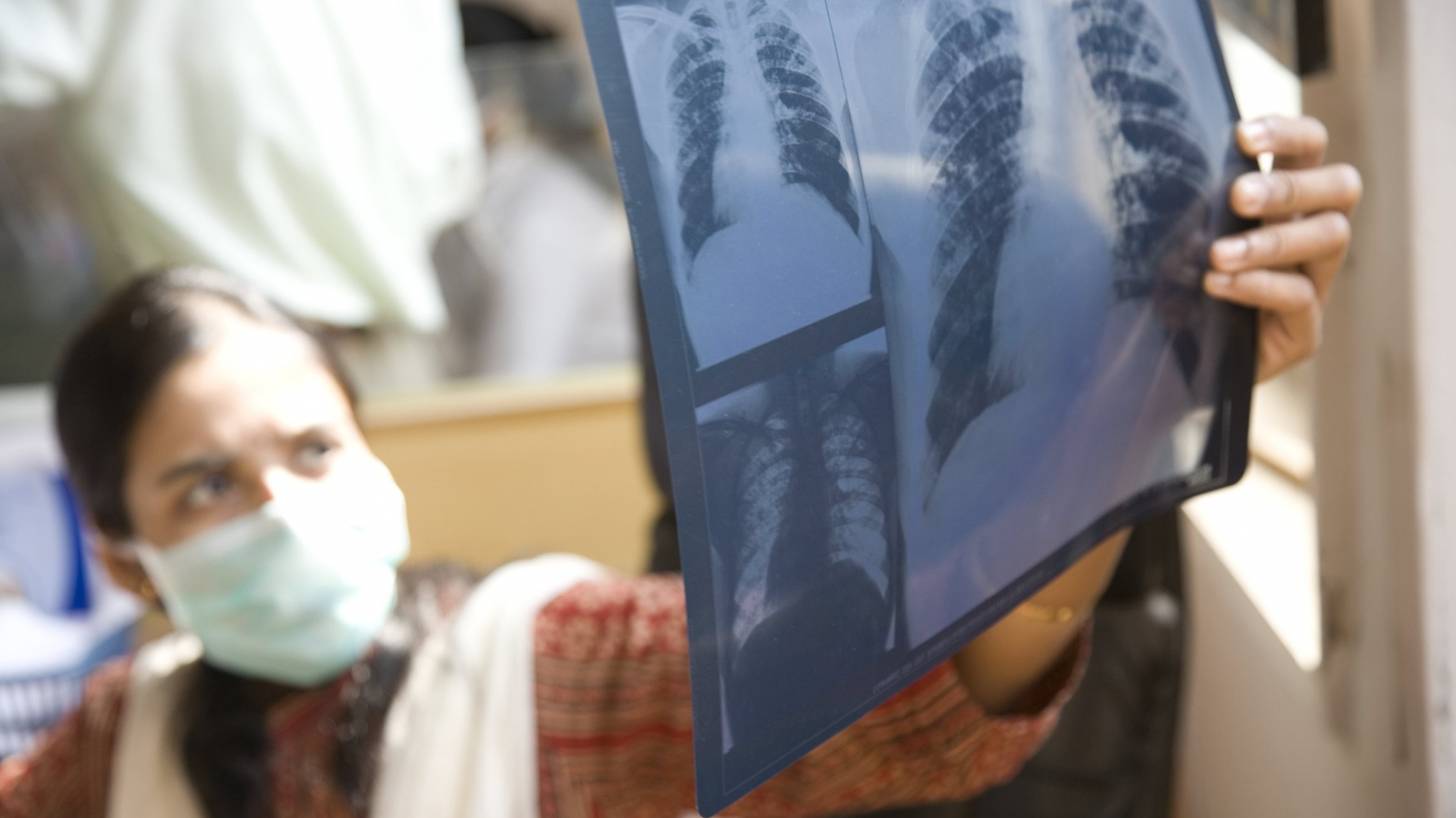
After twenty years of investment in research, tuberculosis (TB) vaccine development has strengthened, with sixteen candidates with the potential to help reach the global TB elimination target in 2030.
Announced today, Wellcome and the Bill & Melinda Gates Foundation are funding $550 million to advance the TB vaccine candidate M72/AS01E (M72) through a Phase III clinical trial.
If proven effective, M72 could become the first vaccine to help prevent pulmonary TB, a form of active TB, in more than 100 years.
Over the past century, the Mycobacterium Bovis Bacille Calmette-Guerin (BCG) vaccine has been deployed worldwide. And various BCG versions are in use in 2023.
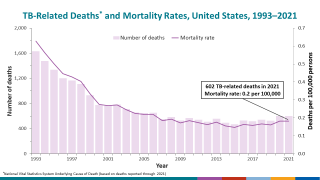
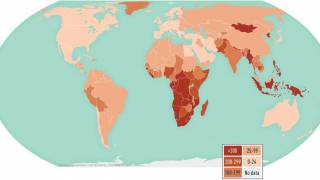
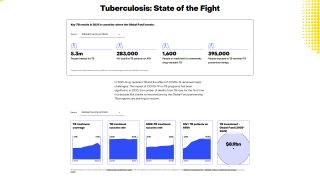
In 2021, an estimated 10.6 million people fell ill with TB, and 1.6 million died, primarily in low- and middle-income countries.
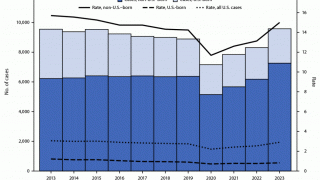
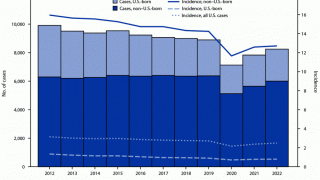
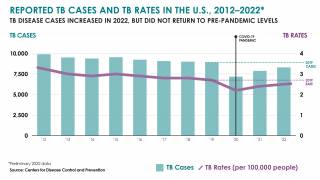
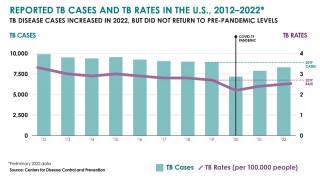
In the U.S., the Centers for Disease Control and Prevention (CDC) reported on March 23, 2023, TB cases increased by 5% in 2022, with 60 U.S. states, the District of Columbia, and territories provisionally reporting 8,300 TB cases.
And about 13 million people live with latent TB infection in the U.S., in states such as Texas, California, and New York.
To achieve the 2030 United Nations Sustainable Development Goals target of an 80% reduction in the TB incidence rate compared with 2015, the world urgently needs new tools to combat this disease.
“With TB cases and deaths on the rise, the need for new tools has never been more urgent,” said Bill Gates, co-chair of the Bill & Melinda Gates Foundation, in a press release on June 28, 2023.
“Greater investment in safe and effective TB vaccines alongside new diagnostics and treatments could transform TB care for millions of people, saving lives and lowering the burden of this devastating and costly disease.”
The Phase III clinical trial will be conducted in collaboration with an international consortium of TB clinical investigators and will enroll approximately 26,000 people, including people living with HIV and without TB infection, at more than 50 trial sites in Africa and Southeast Asia.
The Gates MRI will announce additional details about the trial design and participants in the coming months.
WHO Director-General Dr. Tedros Adhanom Ghebreyesus said the support by the Gates Foundation and Wellcome to develop a new TB vaccine, and the opportunity of September’s UN high-level meeting on TB, shows the world can turn the tide on the TB crisis through sustained political and financial action.
“The tuberculosis crisis demands a new vaccine to reduce disease transmission and avoidable death, especially targeting adults and adolescents who carry at least 90% of the TB epidemic’s burden,” commented Dr. Tedros.
To support the M72 Phase III clinical trial, which will cost an estimated US$550 million, Wellcome is providing up to US$150 million, and the Gates Foundation will fund the remainder, about US$400 million.
Our Trust Standards: Medical Advisory Committee