Old Tuberculosis Vaccine Gets Energized

While versions of the Bacille Calmette-Guerin (BCG) vaccine have been used successfully for decades, researchers have pursued newer vaccines based on innovative technologies.
Today, the Access to Advanced Health Institute (AAHI) published the results of an early-stage clinical trial of a new vaccine candidate targeting tuberculosis (TB), the world's second-deadliest infectious disease.
AAHI's TB vaccine candidate combines several proteins from Mycobacterium tuberculosis (Mtb), the bacterium that causes TB, into a fusion protein ("ID93") with a proprietary immune-stimulating adjuvant ("GLA-SE").
This single-vial TB vaccine is a freeze-dried formulation that can be stored at elevated temperatures (nearly 100 degrees Fahrenheit) for several months, essential for use in developing countries.
AAHI stated on March 6, 2023, the novel single-vial presentation of this freeze-dried TB vaccine candidate elicited a more robust immune response than administration of the same vaccine using separate vials of antigen and liquid adjuvant formulation.
Notably, the thermostable single-vial presentation induced higher levels of antibodies than the two-vial presentation while retaining the vaccine candidate's ability to activate "helper T cells" that recruit other immune cells for a more robust immune response.
"Adjuvanted subunit vaccines have re-energized the field of TB vaccine development. This study represents the first temperature-stable adjuvant-containing subunit TB vaccine candidate to be evaluated in the clinic," says Christopher Fox, Ph.D., Senior Vice President of Formulations and Principal Investigator of the contract awarded by the National Institutes of Health that funded the trial.
"An effective thermostable TB vaccine would not only be better suited to reach areas of the world most burdened by the disease, but it would also mitigate costs and reduce wastage associated with more stringent cold-chain storage requirements," added Fox in a press release.
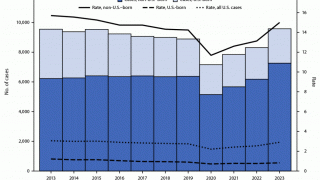
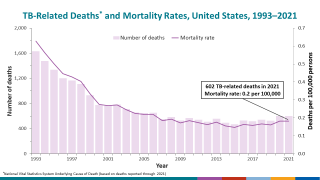
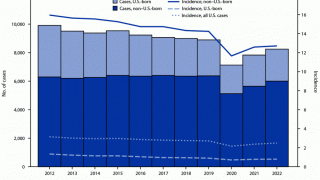
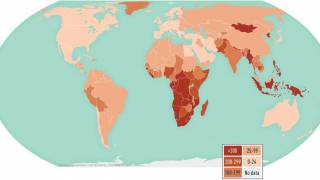
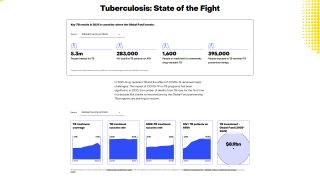
TB spreads when infected people expel the bacterium into the air, says the U.S. Centers for Disease Control and Prevention (CDC). Up to 13 million people in the United States live with latent TB infection.
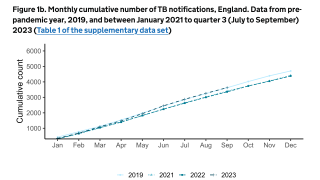
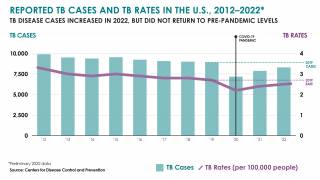
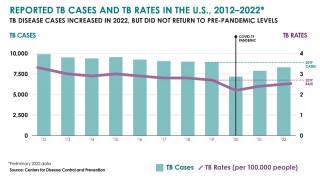
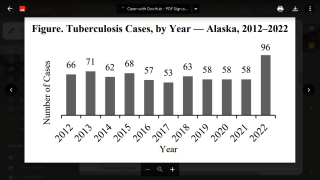
In 2021, the CDC reported 7,882 TB cases from 60 U.S. jurisdictions.
Recent data indicates TB rates are accelerating in certain cities, such as Dallas and Houston.
"The global laboratory-supported systems and networks are critical for detecting and monitoring HIV and tuberculosis," commented Dr. Hank Tomlinson, Director of CDC's Division of Global HIV and TB, in a press statement on February 28, 2023.
Additionally, the BCG vaccine has been deployed against certain types of bladder cancer.
As of March 6, 2023, various BCG vaccine versions are used globally.
However, access to the BCG vaccine is limited in the U.S.
Our Trust Standards: Medical Advisory Committee