Include HIV Patients in TB Vaccine Studies

The peer-review journal The Lancet HIV recently published the results of an extensive study by vaccine experts that concluded HIV patients must be included in clinical trials for new tuberculosis (TB) vaccine candidates currently in development.
And ensure people living with HIV have access to safe and effective TB vaccines, similar to the general population.
This recommendation is essential since people with HIV, at high risk of acquiring Mycobacterium tuberculosis infection and tuberculosis disease, tend to develop less robust vaccine-induced immune responses, wrote these experts on October 11, 2022.
These experts were convened by the DAIDS Cross-Network TB Vaccine Working Group comprised of the HIV Vaccine Trials Network (HVTN), the AIDS Clinical Trials Group (ACTG), and the International Maternal Pediatric Adolescent AIDS Clinical Trials Network (IMPAACT).
This research noted that people living with HIV have a 15- to 21-fold more significant likelihood of developing TB disease and dying from it compared with those not living with HIV.
"People living with HIV are at high risk for tuberculosis infection and subsequent disease and tend to develop less robust vaccine-induced immune responses," said James G. Kublin, MD, executive director of HVTN, based at Fred Hutchinson Cancer Center and co-senior author of the paper.
Moreover, people living with HIV historically have been excluded from TB vaccine trials so developers could maximize the ability to demonstrate strong immunity and effectiveness with their vaccine candidates, Kublin explained.
"Yet, many questions remain unanswered about developing an effective TB vaccine for this group," added Kublin in a related press release.
While the current Bacillus Calmette-Guérin (BCG) vaccine has been used for various diseases over the past 100 years, no U.S. FDA-approved HIV vaccine is available as of October 15, 2022.
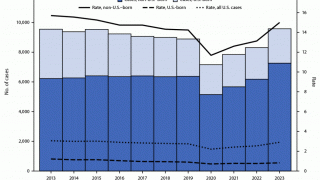
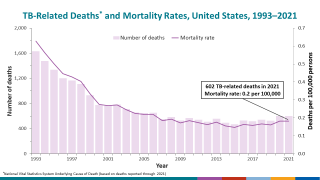
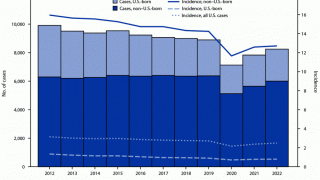
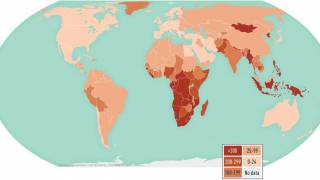
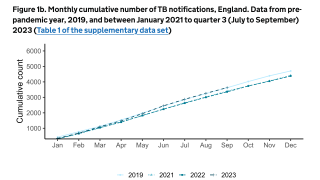
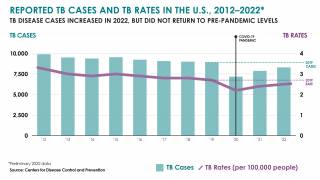
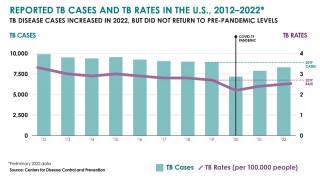
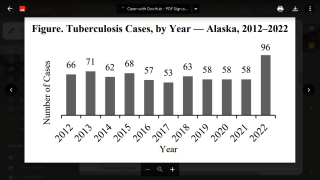
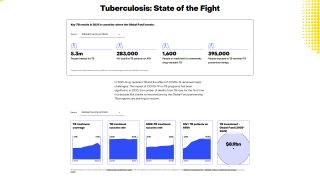
Tuberculosis was responsible for 1.5 million deaths in 2020 and continues to threaten global health.
The World Health Organization estimated that almost 10 million people developed the disease in 2020, 8% of whom were coinfected with HIV.
That data translates to almost 800,000 diagnosed cases that caused 214,000 deaths among people living with HIV.
"It is critical that persons living with HIV across the lifespan be included in TB vaccine trials," commented Amita Gupta, MD, MHS, FIDSA, director of the Division of Infectious Diseases at Johns Hopkins School of Medicine, vice-chair of the IMPAACT TB Scientific Committee and a study co-author.
"Data for children, adolescents and pregnant women are much-needed but are often slow to be generated."
"The goals of TB elimination require novel TB vaccines, and we must focus on these populations who are at especially high risk for TB disease after exposure," Gupta added.
This international expert panel formulated consensus statements with the intention of informing tuberculosis vaccine development and the prioritization of clinical trials for the inclusion of people with HIV.
Additional HIV vaccine candidates and monoclonal antibody development news are posted at PrecisionVaccinations.com/HIV.
HVTN is an international collaborative based at the Fred Hutchinson Cancer Center in Seattle to facilitate the evaluation of vaccines to prevent HIV/AIDS, tuberculosis, and COVID-19. It is the world's most extensive publicly-funded international collaboration facilitating the evaluation of vaccines to prevent HIV/AIDS.
PrecisionVaccinations publishes fact-checked, research-based vaccine news manually curated for mobile readership.
Our Trust Standards: Medical Advisory Committee