Either Td or Tdap Vaccines Are Safe
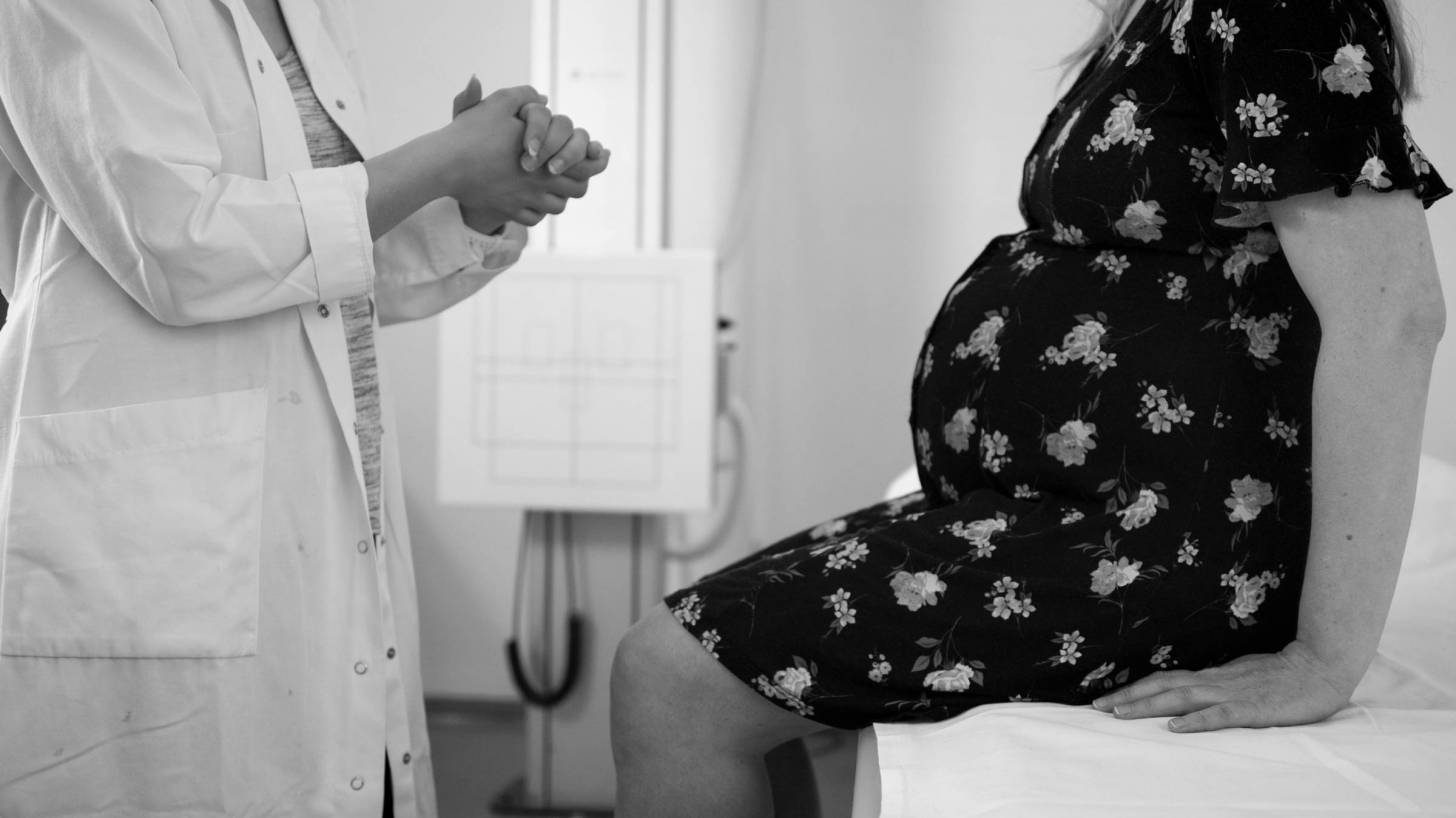
The Advisory Committee on Immunization Practices (ACIP) voted unanimously to recommend that when the Td vaccine (tetanus and Diptheria) is indicated the Tdap (tetanus, Diptheria and pertussis) vaccine is an acceptable option in most situations.
Heidi Splete with MDedge News reported from the Centers for Disease Control and Prevention (CDC)’s meeting on October 23, 2019, that the ACIP members agreed that current data support the use of either the Tdap or Td vaccine in three areas:
- as a decennial booster
- for tetanus prophylaxis and in the setting of wound management
- for additional catch-up doses based on the immunization schedule for persons aged 7 years and older
Additionally, ‘the safety data showed no differences between Tdap and Td, including data regarding pregnant women,’ said Fiona Havers, M.D., of the CDC’s National Center for Immunization and Respiratory Diseases.
The ACIP’s revised language advises that booster doses of “either Td or Tdap” every 10 years throughout life are recommended for continued protection against tetanus and diphtheria.
In addition, either Td or Tdap should be used if a tetanus toxoid–containing vaccine is indicated for prophylaxis in nonpregnant individuals.
For catch-up recommendations, which also apply to pregnant women, the ACIP committee approved the following wording for a series of 3-doses for individuals aged 7-18 years and 19 years and older who have never been vaccinated, that “the preferred schedule is a dose of Tdap (preferably the first dose), followed by either Tdap or Td at least 4 weeks afterward and another dose of either Td or Tdap 6-12 months later.’
‘Individuals in these same age groups who are not fully vaccinated should receive one dose of Tdap, and a dose of either Td or Tdap, if additional doses are needed.’
Previously, the CDC’s MMWR reported a study’s findings on October 18, 2019, that highlighted influenza and pertussis disease burden among pregnant women and infants and vaccination coverage among pregnant women in the USA, and suggest that disease burden could be reduced by improving vaccination coverage.
Many pregnant women do not receive both vaccines recommended during pregnancy, increasing their and their newborns’ risk for influenza and pertussis infection and their potentially devastating consequences.
This study found maternal influenza and Tdap vaccination coverage rates reported as of April 2019 were 53.7 and 54.9 percent, respectively.
Among women whose healthcare providers offered vaccination or provided referrals, 65.7 percent received the influenza vaccine and 70.5 percent received the Tdap vaccine.
The most commonly reported reasons for non-vaccination were believing the vaccine is not effective (influenza; 17.6%) and not knowing that vaccination is needed during each pregnancy (Tdap; 37.9%), followed by safety concerns for the infant (influenza =15.9%; Tdap = 17.1%).
Although pregnant women differ in responses to vaccination offers and referrals, healthcare providers remain their most trusted source of vaccine information, said the CDC.
Starting maternal vaccination discussions with patients early in pregnancy can offer providers multiple opportunities to share information tailored to individual patients’ needs and address vaccination-related concerns.
Recommendations made by the ACIP are not considered final until they are published in the Morbidity and Mortality Weekly Report.
The CDC’s ACIP members had no financial conflicts to disclose.
Vaccine news published by Precision Vaccinations
Our Trust Standards: Medical Advisory Committee