MERS Vaccine Candidate Showed Positive Phase 1 Results

A pharmaceutical company announced positive Phase 1 results of its collaborative vaccine study with INO-4700 (GLS-5300) against Middle East Respiratory Syndrome (MERS).
This MERS study, in partnership with the Walter Reed Army Institute, showed that the vaccine was well-tolerated and demonstrated overall high levels of antibody responses in roughly 95% of subjects.
Additionally, Inovio Pharmaceuticals, Inc.’s INO-4700 generated broad-based T cell responses in nearly 90% of study participants.
In preclinical testing, INO-4700 induced 100% protection from a live virus challenge in a rhesus macaque, non-human, primate study.
In collaboration with GeneOne Life Science, Inovio plans to begin a Phase 1/2 study for MERS in the third quarter of 2018. The study will be conducted by GeneOne Life Science in Korea and fully funded by a $34 million grant from the Samsung Foundation through the International Vaccine Institute.
MERS is caused by a coronavirus that is related to the virus which causes severe acute respiratory syndrome (SARS).
While the SARS coronavirus infection caused illness in more than 8,000 people worldwide, the disease was short-lived between 2002 and 2004 and had a case fatality rate of about 10%.
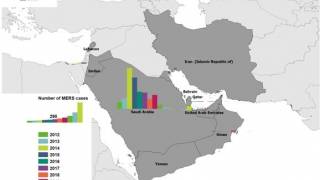
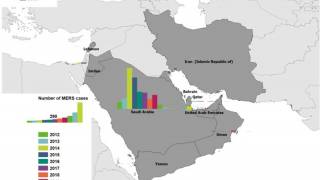
Since the MERS-CoV was first identified in Saudi Arabia in 2012, as of May 2018 the World Health Organization indicates that laboratory-confirmed MERS cases have been reported for 2,220 people worldwide, with 790 deaths, for a case fatality rate of 36%.
Local occasional transmission is still ongoing, primarily in Saudi Arabia where a hospital outbreak occurred earlier this year.
Highlighting the global concern for MERS, in the summer of 2015 a single business person returned to South Korea from Saudi Arabia and was the index case for a South Korean epidemic in 17 hospitals around the country.
That epidemic was comprised of 186 confirmed cases with a 20% case fatality rate.
In April 2018, Inovio was awarded $56 million to develop a MERS vaccine through Phase 2 by The Coalition for Epidemic Preparedness Innovations (CEPI).
The shared goal of Inovio and CEPI is for the MERS vaccine to be available for stockpile as soon as possible for emergency use. The CEPI funding also included support for Inovio’s vaccine against the Lassa virus.
Inovio is a late-stage biotechnology company focused on the discovery, development, and commercialization of DNA immunotherapies that transform the treatment of cancer and infectious diseases.
Our Trust Standards: Medical Advisory Committee