MERS Antibody Treatment Clears Phase 1 Clinical Trial

Recent news regarding the intercontinental transmission of Middle East Respiratory Syndrome (MERS), highlights the global threat posed by emerging infectious diseases, in an increasingly connected world.
MERS is a coronavirus, first identified in Saudi Arabia in 2012, and is highly contagious in healthcare settings.
Most people infected with MERS developed a severe acute respiratory illness, including fever, cough, and shortness of breath. MERS has a case-fatality rate of approximately 35%.
Currently, there are no treatments or vaccine for MERS.
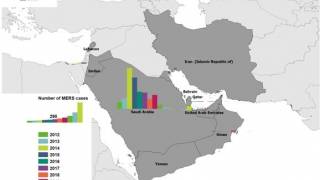
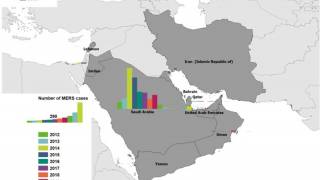
The countries with confirmed MERS cases:
- Bahrain, Iran, Jordan, Kuwait, Lebanon, Oman, Qatar, Saudi Arabia, United Arab Emirates (UAE), and Yemen.
Countries with reported travel-associated MERS cases:
- Algeria, Austria, China, Egypt, France, Germany, Greece, Italy, Malaysia, Netherlands, Philippines, Republic of Korea, Thailand, Tunisia, Turkey, United Kingdom, and United States of America.
In a small phase 1 clinical trial, researchers reported the safety of a fully human polyclonal IgG antibody (SAB-301) produced from the hyperimmune plasma of transchromosomic cattle immunized with a MERS coronavirus vaccine, according to a study published in The Lancet Infectious Diseases.
This is the first time cattle antibodies have been harvested to create a treatment for MERS. SAB-301 uses cattle that have been genetically altered to contain human antibodies to MERS-CoV, the MERS virus.
Using cattle allows researchers to gather a large number of antibodies and is a relatively quick process, with the polyclonal therapy taking only about 3 months to manufacture. This novel treatment for MERS-CoV used cattle antibodies to treat the virus in humans has shown to be safe and well-tolerated.
John Beigel, MD, Leidos Biomedical Research, and the lead author of the study said, "There are limits for a disease like MERS in getting enough plasma from humans to develop an antibody treatment, so to see this do well was a positive."
"That's a very quick timeline for any sort of antibody therapy," said Dr. Beigel. “The treatment had a long half-life of 28 days.”
In the study, 28 healthy volunteers received a single injection of the antibody therapy in doses ranging from 1 milligram per kilogram (mg/kg) to 50 mg/kg. About 20% of study participants reported a headache, while 11% had symptoms similar to the common cold, which can be caused by coronaviruses.
All subjects who received a dose of the antibody treatment showed anti-MERS neutralizing antibody titers that correlated with serum SAB-301 concentrations.
Beigel said that the next step for SAB-301 is to test the treatment in study populations faced with MERS, likely in Saudi Arabia.
And if the treatment proves successful, it could mean transchromosomic cattle, and their antibodies could be used to treat other infectious diseases, including severe acute respiratory syndrome (SARS), Ebola, and pandemic influenza.
"Our study is noteworthy by showing the potential safety of a novel production platform that can quickly generate a new putative drug candidate to an emerging infectious disease," Beigel and his co-authors concluded in the study.
This phase 1 clinical trial was conducted by the National Institute of Health's National Institute of Allergy and Infectious Diseases (NIAID).
Our Trust Standards: Medical Advisory Committee