Dengue Vaccination Pre-Testing Reaffirmed
Prior to receiving the Dengvaxia dengue vaccine, health officials continue to recommend individual testing to determine if each person has had a previous dengue virus infection.
“Dengvaxia vaccination is only appropriate for those who have had a previous, specific type of dengue infection may be counterintuitive," said the World Health Organization (WHO).
There are 4 distinct, but closely related, serotypes of the virus that cause dengue; DEN-1, DEN-2, DEN-3, and DEN-4. Recovery from infection by one dengue virus provides lifelong immunity against that particular serotype.
This WHO recommendation was published in a position paper on September 7, 2018.
The new WHO position paper replaces its 1st publication regarding Dengvaxia, the first dengue vaccine approved for use in 2016.
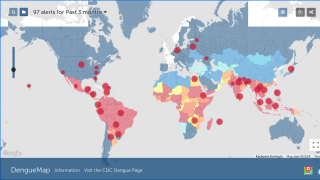
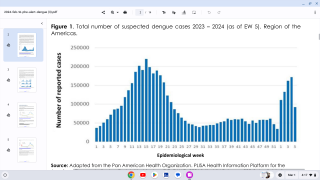
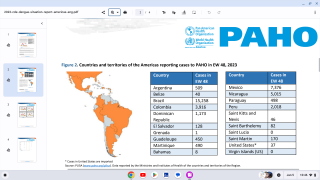
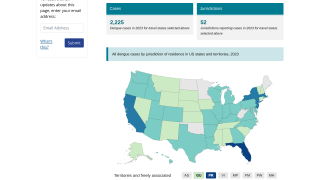
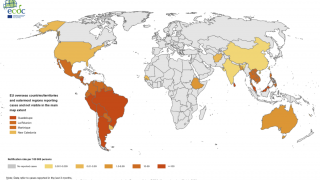
This is an important recommendation since dengue virus outbreaks continue to be reported worldwide. Approximately 50 million new dengue infections are estimated to occur annually, in more than 100 endemic countries, says the WHO.
Recently, countries such as Thailand and France’s Reunion Island have reported significant dengue outbreaks.
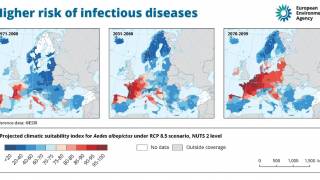
Dengue is a mosquito-borne viral disease that has rapidly spread in all regions of WHO in recent years. Dengue virus is transmitted by female mosquitoes mainly of the species Aedes aegypti and, to a lesser extent, Ae. albopictus, which also transmits chikungunya, yellow fever, and Zika viruses.
Dengue is found in tropical and sub-tropical climates worldwide, mostly in urban and semi-urban areas.
In late 2015 and early 2016, the first dengue vaccine, Dengvaxia (CYD-TDV) by Sanofi Pasteur, was registered in several countries for use in individuals 9-45 years of age, living in endemic areas.
But, during April 2018, the World Health Organisation (WHO) Strategic Advisory Group of Experts (SAGE) on Immunization said the Dengvaxia vaccine should be managed much more carefully.
SAGE said that it would be unsafe to give the Sanofi vaccine to people who had not previously been infected with dengue.
This SAGE recommendation is based upon a 2018 study, published in the journal Science, found there are 4 species of dengue virus, and infection with 1 of the species does not provide protection from another.
The antibodies from the first infection may cause the second infection to be more serious. Which means, the antibodies actually backfire, causing harm to the patient.
Currently, there is another dengue vaccine candidate in Phase 3 clinical trials, Takeda’s TAK-003.
Vaccines for international travelers are available at certified pharmacies in the USA.
** Schedule travel vaccine appointment here **
Vaccines, like any medicine, can have side effects. You are encouraged to report negative side effects of vaccines to the FDA or CDC.
Our Trust Standards: Medical Advisory Committee