Texas Biomed Partners With Marburg Virus Vaccine Candidate

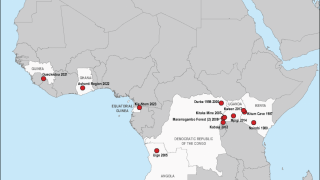
First identified in Germany in 1967, Marburg virus (MARV) outbreaks have been reported more than a dozen times over the past 56 years.
A Marburg virus (MARV) vaccine tested at Texas Biomedical Research Institute is progressing in clinical trials, moving a step closer to becoming the world’s first vaccine against the deadly virus.
The Sabin Vaccine Institute recently announced it launched Phase 2 clinical trials of its single-dose vaccine candidate ChAd3-MARV.
Early tests demonstrating the vaccine’s efficacy, safety, and optimal dosage were completed at Texas Biomed.
This announcement is essential since there are no approved Marburg vaccines or treatments.
“We have been partnering with Sabin since 2019 and are very excited to see their Marburg vaccine candidate move into Phase 2 clinical trials,” says Ricardo Carrion, Jr., Ph.D., the Director of Maximum Containment Contract Research at Texas Biomed, in a press release on December 7, 2023.
“An effective vaccine is critical to protect people from this deadly virus, especially as we see the frequency of outbreaks increasing in more places.”
The Phase 2 clinical trial will build on promising results from preclinical studies and a smaller Phase 1 clinical trial.
Texas Biomed continues to partner with Sabin to gather more detailed information that can only be gained through tightly controlled animal studies, including how soon protection is induced after vaccination.
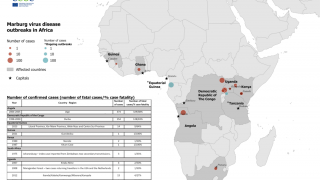
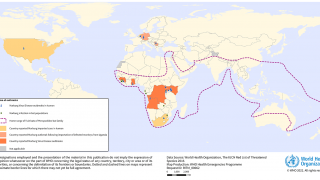
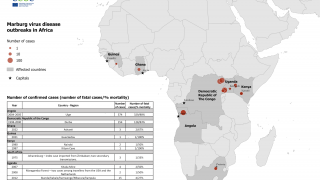
Marburg virus is a part of the same filovirus family as Ebola virus and causes severe hemorrhagic fever. It is incredibly deadly, with up to a 90% fatality rate.
Recent MARV outbreaks that occurred in Equatorial Guinea killed 12 out of 17 confirmed cases, with another 23 probable deaths, according to the World Health Organization.
Tanzania also saw its first-ever Marburg outbreak, which killed six out of eight confirmed cases.
Texas Biomed has conducted similar work on Sabin’s closely related Sudan ebolavirus vaccine as part of a World Health Organization-coordinated outbreak response.
As of December 24, 2023, several Ebola vaccines, therapies, and vaccine candidates are conducting research studies. Ebola vaccines are not generally available in the U.S.
Our Trust Standards: Medical Advisory Committee