TB Vaccine Accelerator Council Supports Novel Tuberculosis Vaccine Development

The World Health Organization (WHO) Director-General, Dr. Tedros Adhanom Ghebreyesus, recently launched the TB Vaccine Accelerator Council to facilitate the development, licensing, and using new Tuberculosis (TB) vaccines.
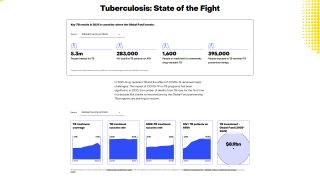
The 100-year-old Bacille Calmette-Guérin (BCG) is the only licensed TB vaccine, with over ten versions available globally.
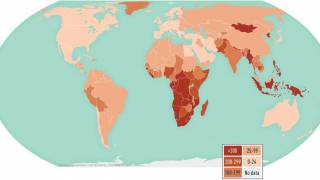
While it provides moderate efficacy in preventing severe forms of TB in infants and young children, it does not adequately protect adolescents and adults, who account for the majority (>90%) of TB transmission globally, said the WHO on September 22, 2023.
In a press release, Dr. Tedros Adhanom Ghebreyesus commented, "Today, we have knowledge and tools they could only have dreamed of."
"The political declaration countries approved today, and the targets they have set, are a commitment to use those tools, and develop new ones, to write the final chapter in the story of TB."
The Council aims to identify innovative sustainable financing, market solutions, and partnerships across public, private, and philanthropic sectors. It will leverage various agencies to strengthen commitment and actions for novel TB vaccine development and access.
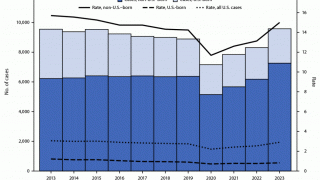
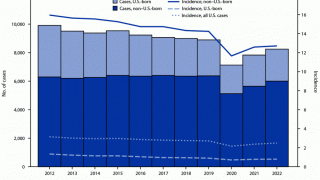
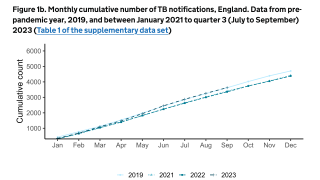
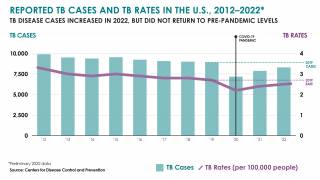
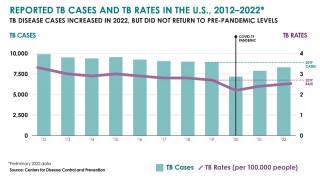
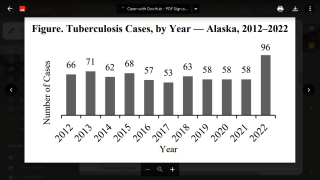
The U.S. Centers for Disease Control and Prevention (CDC) reported that TB outbreaks increased by 5% in 2022, with 60 U.S. states, the District of Columbia, and territories provisionally reporting 8,300 TB cases last year.
According to the CDC, TB is caused by Mycobacterium tuberculosis. The bacteria usually attack the lungs, but TB bacteria can attack any body part.
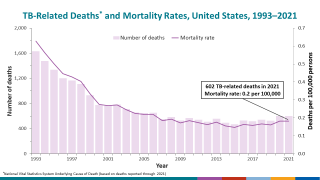
As a result, two TB-related conditions exist: latent TB infection and TB disease. If not treated properly, TB disease can be fatal.
In the U.S., Merck's TICE BCG vaccine has limited availability.
Our Trust Standards: Medical Advisory Committee