How Many Malaria Vaccinations Are Needed

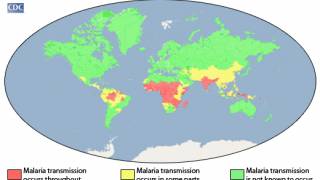
Over the past two years, malaria has become a vaccine-preventable disease thanks to the approval of two innovative vaccines.
The World Health Organization (WHO) now recommends that children in certain areas receive four doses of GSK's Mosquirix™ (RTS,S/AS01) malaria vaccine, the world's first approved malaria vaccine.
As of May 2024, Mosquirix is available in African countries but not in the United States.
Because many malaria infections are asymptomatic, researchers conducted a large-scale parasite genotyping phase 2b clinical trial to explore how the vaccine dosing regimen affects efficacy.
These researchers concluded that all tested dosing regimens similarly blocked some infections. This finding, published by The Lancet Infectious Diseases on May 6, 2024, could suggest new strategies for future malaria vaccine development and implementation.
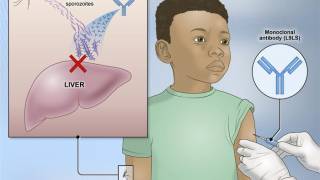
RTS,S is a pre-erythrocytic vaccine based on the Asn-Ala-Asn-Pro repeat and C-terminal regions of the most abundant antigen on the sporozoite surface, the circumsporozoite protein. It is fused with the hepatitis B virus surface antigen and formulated with a liposome-based adjuvant, AS01.
In The Lancet Infectious Diseases, Daniel Neafsey and colleagues conducted a large longitudinal parasite genotyping study (MAL-095) from two follow-up periods of a phase 2b open-label randomized controlled trial (MAL-094).
The MAL-094 trial tested alternative dosing schedules of RTS,S based on earlier studies in malaria-naive adults that showed higher efficacy when, in a three-dose regimen, the last dose was fractionated to a fifth of the standard dose.
Genotyping indicated that RTS,S vaccination can block infections before the parasite can reach the blood stage.
In 1,500 children aged 5–17 months in Ghana and Kenya, vaccine efficacy against clinical malaria was similar using the standard three total doses or the fractional third dose regimen from either month two or month 7, suggesting flexibility in dosing regimens and intervals.
To better understand the mechanism of RTS,S protective efficacy, Neafsey and colleagues examined asymptomatic infections as vaccine efficacy outcomes.
Genotyping was used to study the time to the first new malaria infection and the number of new malaria infections acquired over time for two follow-up periods.
The molecular detection of new infections as an outcome was done.
The parasite positivity rate by molecular detection was about 3% higher than by microscopic identification (14% [5078/35 456] vs. 11.6% [4115/35 456]).
Notably, there was no difference in protective efficacy with head-to-head comparisons of RTS,S dosing regimens (per protocol set: 37–43%, 95% CI 21–53; exposed set: 25–31%, 9–43%) and the efficacy was significantly different from the control group (p<0·0033).
A full dose at month two did appear to provide more sustained protection than a fractional dose at month 2.
Compared with the control group, the groups that received the RTS,S vaccines had a lower number of new infections (e.g., 2·6–3 vs. 4.1), had reduced risk of polyclonal first infections (p<0·0001), and had a reduction in the maximum number of distinct haplotypes detected in a sample.
The unexpected positive association between infection status at the first vaccination and vaccine efficacy was reported, but future studies are needed to substantiate this association.
Likewise, analyzing over 36,000 samples, there appears to be no difference in vaccine efficacy against the first new infection with 3D7-matched or mismatched strains.
However, individual data suggest that mismatches at specific amino acid residues in the 3D7 circumsporozoite C-terminus (e.g., at amino acid positions 322, 324, and 327) could have resulted in differential vaccine efficacy.
The difficulty in matching haplotypes with vaccine efficacy could also be attributed to the diversity of the circumsporozoite gene. A larger dataset might provide clues for structure-based design for effective vaccine-induced immunity.
Further immune assays to understand the mechanism of the observed protective efficacy could lead to more effective, longer-lasting malaria vaccines.
Funding for this study was provided by GlaxoSmithKline Biologicals SA, PATH, Bill & Melinda Gates Foundation, and the German Federal Ministry of Education and Research.
In a separate study published by The Lancet in April 2024, three primary doses were effectively deployed in African countries in the phase 3 trial. There was no evidence of the safety signals observed, and introducing the vaccine was associated with substantial reductions in hospital admissions with severe malaria.
Our Trust Standards: Medical Advisory Committee