DogBot Explains Which Dogs Can Fly to the United States

While rabies was officially eliminated in dogs in the United States in 2007, unvaccinated canines can still contract the deadly disease from rabid wildlife, including bats.
If you plan on bringing a dog into the U.S. this summer, new rules from the government may impact your plans.
The U.S. Centers for Disease Control and Prevention (CDC) announced on May 8, 2024, new dog importation rules will be implemented in August 2024.
The CDC stated that international travelers should use the DogBot to determine which rules are applicable based on the travel dates and where their dog travels.
These rules apply to all dogs, including puppies, service animals, and dogs that left the U.S. and are returning. They also apply whether you are a U.S. citizen, legal U.S. resident, or foreign national.
If you don't follow the CDC's new rules, your dog will not be allowed to enter the U.S.
According to the CDC, if a country or political unit is not listed, it is not classified as high risk for importing dog rabies into the United States. Therefore, if the country is not listed, the CDC strongly recommends vaccinating against rabies. However, the additional requirements for dogs from high-risk countries to enter the United States do not apply.
To learn how these rules apply to your dog, please visit the "Bringing a Dog into the United States" website.
Previously, the U.S. Transportation Security Administration advised how pets should be brought through airport security checkpoints.
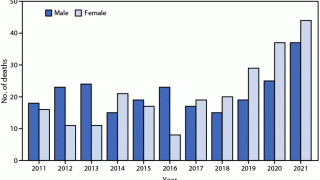
Over the past few years, bats have become the leading source of rabies in people in over 40 U.S. states. The CDC says about 1-3 rabies cases are confirmed annually.
However, about 60,000 Americans get post-exposure prophylaxis each year to prevent rabies infection after being bitten or scratched by an infected or suspected infected animal.
Various rabies vaccines have been U.S. FDA-approved for decades. However, a next-generation rabies vaccine could soon replace current options.
In April 2024, YS Biopharma Co., Ltd. announced positive results from the ongoing Phase 3 clinical trial of its PIKA Rabies Vaccine.
This vaccine can potentially meet the goal of a one-week regimen to replace the conventional three- or four-week regimens.
PIKA was granted U.S. FDA orphan drug designation for prevention of rabies virus infection, including post-exposure prophylaxis for rabies.
Our Trust Standards: Medical Advisory Committee