Yale Researchers Patent Next-Gen Malaria Vaccine Candidate

A Yale researcher has partnered with Novartis to develop an RNA vaccine to protect against the malaria parasite, reported Yale News.
According to the Yale research team, if the Malaria vaccine candidate is approved, it could be revolutionary to the pharmaceutical field.
The RTS,S vaccine, which was developed about two and a half years ago, grants about 30% protection, which decreases very quickly in about two to three years, said this Yale article written by Beatriz Horta.
The vaccine patent, originally applied for in 2014 by Yale researchers, was published by the U.S. Patent & Trademark office this year on February 4, 2021.
The Yale School of Medicine Chief of Rheumatology, Allergy and Immunology Richard Bucala, M.D., Ph.D., ’79 and his lab used self-amplifying RNA (saRNA) technology to improve the effectiveness and speed of production of the vaccine by decreasing the amount of genetic material needed in each shot.
sRNAs are pieces of genetic material that contain the code for the protein to be targeted by the body’s antibodies after getting the vaccine but also hold the code for an enzyme that allows the genetic material to replicate itself several times.
This means that a much smaller amount of genetic material needs to be injected for the vaccine to provide adequate immunization.
According to Dr. Bucala, one of the issues with developing a malaria vaccine is that there is no natural sterilizing immunity, which prevents reinfection in the long-term or, ideally, throughout a person’s lifetime.
This means that vaccines developed against malaria will usually provide only a short protection period or provide low immunity.
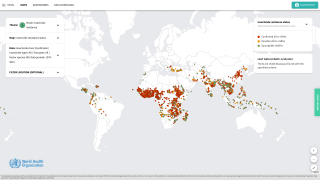
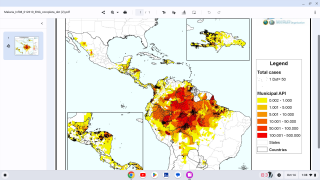
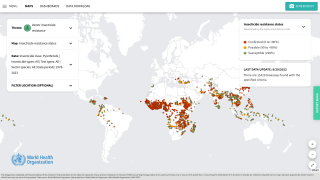
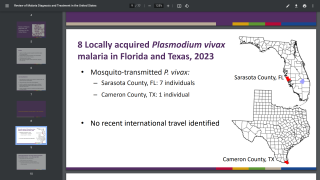
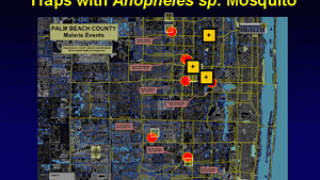
Malaria is a life-threatening disease caused by parasites transmitted to people through the bites of infected female Anopheles mosquitoes. It is preventable and curable.
Also, different from many other vaccine-preventable diseases, malaria is caused by the Plasmodium parasite rather than a virus, which means the organism has a more significant number of genes and has developed more mechanisms to trick the body’s immune system.
“The malaria parasite has evolved elaborate strategies to circumvent destruction by the host immune system, complete its life cycle, and be passed on by mosquitoes to another person so they can be infected,” Bucala added.
The main target of the saRNA vaccine is the parasite’s PMIF protein, which enables the organism to suppress the host’s immune response and prevents it from producing memory T-cells, which generate immunity. By inhibiting the activity of this protein, the vaccine will allow the body to attack the parasite naturally and generate longer-term immunity to the disease.
Bucala’s lab developed the new RNA vaccine in partnership with Andrew Geall, former RNA vaccine platform leader at Novartis Pharmaceuticals.
According to Geall, he had worked previously with Bucala on malaria research and has been interested in developing a vaccine for some time. However, the team faced challenges because of the novelty of the saRNA technology, which was tested for the first time in humans a year ago.
“The main challenge was getting the work published,” Geall wrote in an email to the News. “This is a new concept and the mode of action for the vaccine is very different to the current approaches. The reviewers required extensive data validation.”
According to Geall, the technology behind this vaccine could also significantly boost pharmaceutical companies’ abilities to produce the genetic material and distribute the shots, given the fact that less RNA is required in each dose. This aspect of the vaccine is critical for a disease that infects such a large number of people each year.
“[Malaria is] the second leading cause of infectious disease death in the world last year, about over 400,000 individuals died,” Bucala said. “They’re usually young children under the age of five.”
According to the World Health Organization, children under five account for over 67% of worldwide fatalities related to malaria each year. In 2019, there were an estimated 229 million cases of malaria worldwide.
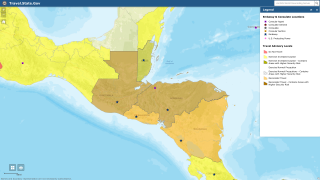
Malaria vaccination is just one of several travel-vaccines suggested by the U.S. CDC when an international traveler visits certain countries. The CDC recommends travelers speak with a travel vaccine specialist one-month before traveling abroad.
PrecisionVaccinations publishes research-based vaccine news.
Our Trust Standards: Medical Advisory Committee