Chikungunya Virus Vaccine Candidate Enters Clinical Study

A biotech company announced the launch of a Phase I clinical trial to evaluate its live-attenuated vaccine candidate VLA1553 against the Chikungunya virus.
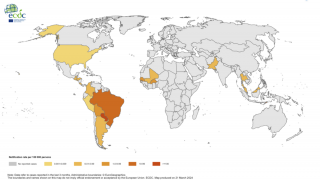
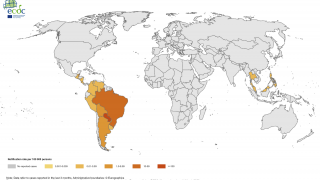
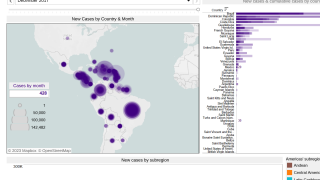
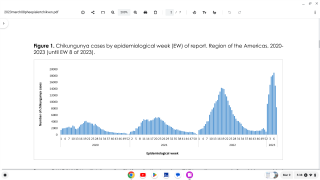
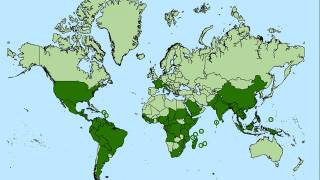
Chikungunya virus (CHIKV) outbreaks have been reported in Asia, Africa, and recently in the Americas.
Since 2013, CHIKV local transmission has been identified in 45 countries or territories throughout the Americas. More than 1.7 million suspected CHIKV cases reported to the Pan American Health Organization from affected areas (Updated data from PAHO).
As of January 9, 2018, a total of 114 chikungunya virus cases have been reported to ArboNET from 26 U.S. states, with 36 additional cases reported from Puerto Rico.
There are no preventive vaccines or effective treatments available and, as such, Chikungunya is considered to be a major public health threat.
Valneva SE is a fully integrated, commercial-stage biotech company focused on developing innovative life-saving vaccines.
The Phase I clinical trial is a randomized, observer-blinded, dose-escalation, multi-center study. It will investigate three different dose levels of VLA1553 in approximately 120 healthy adults vaccinated with a single-shot immunization.
Chikungunya is a mosquito-borne viral disease caused by the Chikungunya virus, a Togaviridae virus, transmitted by Aedes mosquitoes.
Chikungunya virus causes clinical illness in 72-92% of infected humans around 4 to 7 days after an infected mosquito bite. Complications include visual, neurological, heart and gastrointestinal manifestations report the Centers for Disease Control and Prevention (CDC).
Fatalities from CHIKV are seldom reported.
This clinical trial design includes the investigation of antibody persistence and an additional vaccination using the highest dose of VLA1553 at 6 and 12 months, says Valneva SE.
This re-vaccination will serve as an intrinsic human viral challenge, aiming to demonstrate that subjects are protected from vaccine-induced viremia thereby indicating potential efficacy of VLA1553 early in clinical development.
First data from this trial are expected to be available early 2019.
VLA1553 is based on an infectious clone (CHIKV LR2006-OPY1) attenuated by deleting a major part of the gene encoding the non-structural replicate complex protein nsP3, aiming for protection against various Chikungunya virus outbreak phylogroups and strains.
In pre-clinical development, a single-vaccine shot was shown to be highly immunogenic in vaccinated Non-Human Primates (NHP) (cynomolgus macaques) and showed no signs of viremia after challenge, said Valneva SE.
In NHPs, VLA1553 induced a strong, long-lasting (more than 300 days) neutralizing antibody response comparable to wild-type CHIKV infections combined with a good safety profile.
Our Trust Standards: Medical Advisory Committee