Which Students Need Which Meningitis Vaccine?

With Fall classes starting soon, now may be the best time to get those required meningococcal immunizations. Some local schools require seventh and twelfth grade students, as well as most colleges, to provide a shot record before entering class.
To help clarify the meningitis vaccination requirements, the Advisory Committee on Immunization Practices (ACIP) has updated its 2015 recommendations regarding the use of the two meningococcal vaccines.
There are two MenB vaccines licensed by the Food and Drug Administration (FDA) for use in the United States among people aged 10 to 25 years. These two MenB vaccines are not interchangeable, which means the same vaccine must be used for all doses in a vaccination series.
These updated recommendations are regarding use of the 2- and 3-dose schedules of MenB-FHbp vaccine (Trumenba). For persons at increased risk for meningococcal disease and for use during serogroup B meningococcal disease outbreaks, ACIP recommends that 3 doses of MenB-FHbp be administered at 0, 1–2, and 6 months.
When given to healthy adolescents who are not at increased risk for meningococcal disease, the ACIP recommends that 2 doses of MenB-FHbp should be administered at 0 and 6 months.
Additionally, the ACIP recommendations regarding use of MenB-4C vaccine (Bexsero) are unchanged.
The minimum interval between any 2 doses of MenB vaccine is 4 weeks, and may be administered concomitantly with other vaccines. But not at the same location on the body.
The ACIP will consider MenB vaccine booster doses for persons at increased risk for serogroup B meningococcal disease as data becomes available.
As stated in previous ACIP reports on MenB vaccines, vaccination should be deferred in women known to be pregnant or lactating.
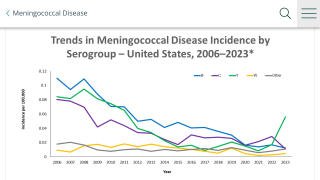
According to the Centers for Disease Control and Prevention (CDC), 10 to 15 percent of people with meningococcal disease die, even with appropriate treatment. Of those who recover, up to 20 percent have serious after effects like permanent hearing loss, limb loss or brain damage.
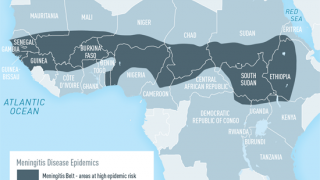
Meningococcal disease can refer to any illness caused by the type of bacteria called Neisseria meningitidis, also known as meningococcus. These illnesses are often severe and can be deadly. They include infections of the lining of the brain and spinal cord (meningitis) and bloodstream infections (bacteremia or septicemia).
These bacteria spread through the exchange of respiratory and throat secretions like spit or kissing.
The symptoms of meningococcal disease are usually sudden onset of fever, headache, and stiff neck. It can start with symptoms similar to influenza (flu), and will often also cause nausea, vomiting, increased sensitivity to light, rash, and confusion.
Keeping up to date with recommended vaccines is the best defense against meningococcal disease.
According to the CDC, there are three types of meningococcal vaccines available in the United States:
- Meningococcal conjugate vaccines (Menactra, Menveo, and MenHibrix)
- Meningococcal polysaccharide vaccine (Menomune)
- Serogroup B meningococcal vaccines (Bexsero® and Trumenba®)
The CDC suggests all 11 to 12 year olds should be vaccinated with a meningococcal conjugate vaccine (Menactra® or Menveo®). A booster dose is recommended at age 16 years. Teens and young adults (16 through 23 year olds) also may be vaccinated with a serogroup B meningococcal vaccine.
In certain situations, other children and adults could be recommended to get any of the three kinds of meningococcal vaccines.
Adverse events occurring after administration of any vaccine should be reported to the Vaccine Adverse Event Reporting System (VAERS), by telephone (1–800–822–7967) or online.
No conflicts of interest were disclosed by these individuals: Patton ME, Stephens D, Moore K, MacNeil JR. Updated Recommendations for Use of MenB-FHbp Serogroup B Meningococcal Vaccine — Advisory Committee on Immunization Practices, 2016. MMWR Morb Mortal Wkly Rep 2017;66:509–513. DOI: http://dx.doi.org/10.15585/mmwr.mm6619a6.
Our Trust Standards: Medical Advisory Committee
- Updated Recommendations for Use of MenB-FHbp Serogroup B Meningococcal Vaccine — Advisory Committee on Immunization Practices, 2
- Meningococcal Disease
- Evidence-Based Recommendations—GRADE
- Role of Factor H Binding Protein in Neisseria meningitidis Virulence and Its Potential as a Vaccine Candidate To Broadly Protect
- Meningococcal Vaccination