Meningococcal B Vaccine 4CMenB Found Safe for Children

Meningococcal disease remains one of the most feared infectious diseases.
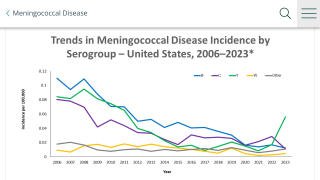
Of the 12 known meningococcal capsular groups, group B (MenB) is responsible for most cases of invasive disease, especially in children.
Unlike the highly effective vaccines against groups A, C, W, and Y, the development of a similar vaccine for MenB has been challenging.
The meningococcal serogroup B vaccine (4CMenB) has been licensed in more than 35 countries.
However, healthcare providers have unanswered questions regarding the fewest number of doses required to induce persistent immune responses.
Researchers did a systematic review and meta-analyses of the 4CMenB vaccine with its originator meningococcal B recombinant vaccine or routine vaccines in children or adolescents.
This meta-analysis was published in the Lancet Infectious Diseases abstract and found the 4CMenB vaccine produced an acceptable short-term safety profile within 30 days of vaccination and seroconversion was 92 percent.
But, a booster dose is required to protect children against strain M10713.
Meningococcal disease is rare, easily misdiagnosed, and potentially deadly. Diagnosis in the early stages is difficult and the disease often progresses extremely rapidly.
Moreover, given the recent outbreak at Oregon State University, where all students under the age of 26 will have to show they've been vaccinated against the Men B strain, vaccination may be the best protective option.
Vaccination is the most effective way to protect against this aggressive disease that has a narrow timeframe for diagnosis and treatment, says the Centers for Disease Control and Prevention (CDC).
Two serogroup B meningococcal vaccines are currently licensed for use in persons aged 10–25 years in the USA.
The two vaccines are MenB-FHbp (Trumenba) and MenB-4C (Bexsero).
Most pharmacies offer MenB vaccines.
The CDC Vaccine Price List provides the private sector vaccine prices for general information. Meningococcal discounts can be found here.
Vaccines, like any medicine, can have side effects, says the CDC. You are encouraged to report negative side effects of vaccines to the U.S. Food and Drug Administration and the CDC.
These researchers did not disclose any conflicts of interest. Correspondence information about the author Prof Lamberto ManzoliEmail the author Prof Lamberto Manzoli, Annalisa Rosso, MD, Carolina Marzuillo, MD, Mario Bergamini, MD, Armando Stefanati, MD, Rosario Cultrera, MD, Prof Paolo Villari, MD, Prof Walter Ricciardi, MD, Prof John P A Ioannidis, MD, Despina G Contopoulos-Ioannidis, MD.
Our Trust Standards: Medical Advisory Committee
- Immunogenicity and safety of the multicomponent meningococcal B vaccine (4CMenB) in children and adolescents: a systematic revie
- Invasive meningococcal disease - Annual Epidemiological Report 2016 [2014 data]
- Meningococcal Carriage Evaluation in Response to a Serogroup B Meningococcal Disease Outbreak and Mass Vaccination Campaign at a
- Hajj-Associated Outbreak Strain of Neisseria meningitidis Serogroup W135: Estimates of the Attack Rate in a Defined Population a
- Updated Recommendations for Use of MenB-FHbp Serogroup B Meningococcal Vaccine — Advisory Committee on Immunization Practices, 2
- A Bivalent Meningococcal B Vaccine in Adolescents and Young Adults