Set-Point Dengue Vaccine Candidate Ready For Human Trials

A UK based biotechnology company announced the successful completion of preclinical testing of its lead vaccine candidate for Dengue Fever, DV1-DV4.
And, the DV1-DV4 vaccine candidate is now transitioning into the human clinical study.
Emergex Vaccines Holding Limited announced in a press release on December 10, 2019, that this new vaccine construct, which comprises Nature’s gene-chip peptides bound to a quantum cluster gold nanoparticle delivery system, was shown to have an excellent safety profile in a repeat dose Good Laboratory Practice (GLP) grade toxicology study using a standard industry model.
No adverse reactions were seen at any dose level during non-human tests.
In addition to providing disease prophylaxis to Dengue Fever, this vaccine candidate may also provide immunity to other flaviviruses including Zika and Yellow Fever, as the peptides chosen in the vaccine construct are highly conserved amongst flaviviruses.
This work builds upon the 2018 preclinical work including GLP toxicology testing of the gold nanoparticle, which will form the base for all of Emergex’s vaccines and efficacy studies of the vaccine candidate.
The gold nanoparticle forms the central core (“scaffold”) for immunogenic peptides that together form the final construct for development, says the company.
The particles can be made to measure less than 5 nanometers in diameter. They act as the carrier system for immunogenic vaccine constructs that comprise synthetic peptides derived from highly conserved components of the disease-causing virus, microbe or pathogen.
These peptides have been reverse-engineered using post-infectious blood to identify and select peptides that elicit the strongest cell-mediated immune response.
Laurens Rademacher, Emergex’s Chief Technology Officer, commented in the press release, “Our vision is to offer affordable, practical, safe and effective vaccine solutions. The success we are reporting today takes us closer to achieving this.”
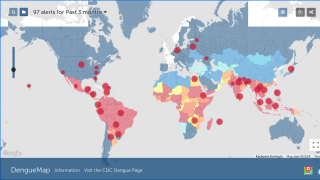
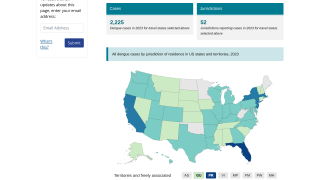
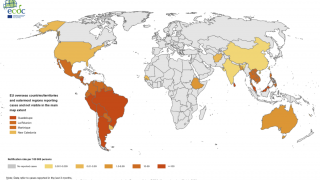
Dengue Fever is a mosquito-borne viral infection prevalent in tropical and semi-tropical parts of the world. In recent decades, Dengue has spread and today approximately half the world’s population is at risk of contracting it.
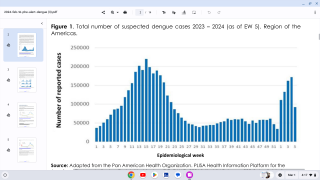
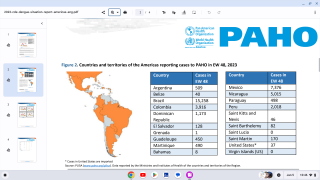
During November, the Pan American Health Organization reported over 2.7 million confirmed Dengue Fever cases so far in 2019.
This is the largest number of Dengue cases recorded in the Americas and has already exceeded by 13 percent the total number of cases reported in 2015, which was the previous record year.
Dengue outbreak news
Dengue is caused by a virus that has 4 different but closely-related serotypes. When a person recovers from the infection, he acquires lifelong immunity against that particular serotype, says the US Centers for Disease Control and Prevention (CDC).
Patients with a Dengue infection usually present with flu-like symptoms, such as a headache and fever.
However, Severe Dengue can occur in up to 5 percent of cases and is a leading cause of hospitalization and death among children in Dengue-affected regions, says the CDC.
The Aedes aegypti female mosquitos are the main source of dengue transmission.
However, new research indicates the Dengue virus can be transmitted during sex between humans.
Regarding preventive vaccines, Dengvaxia is the only approved dengue vaccine in the USA and about 20 other countries. And, there are Dengue vaccine candidates in clinical studies, such as Takeda’s TAK-003.
The CDC says any vaccine can cause side effects, which should be reported to a healthcare provider ASAP.
Emergex Vaccines Holdings Limited is pioneering the development of ‘set-point’ vaccines to address some of the world’s most immediate health threats and serious intra-cellular bacterial infections.
Dengue vaccine news is published by Precision Vaccinations.
Our Trust Standards: Medical Advisory Committee
- Emergex Successfully Completes Preclinical Testing of its Lead ‘Set-Point’ Vaccine Candidate for Dengue Fever and Other Flavivir
- Emergex Vaccines
- Emergex Demonstrates Excellent Safety Profile for Gold Nanoparticle Used in its Vaccine Platform
- Dengue Virus Transmission in Spain Confirmed By Male Partners