Ohio Reaffirms Hepatitis A Virus Outbreak for 2019

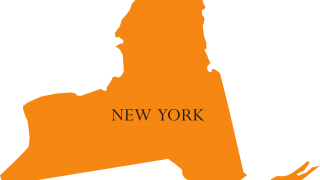
The Ohio Department of Health (ODH) has reaffirmed its statewide community outbreak of the hepatitis A virus for 2019.
As of February 11, 2019, the number of Ohio counties reporting hepatitis A (HepA) cases was 68 (77%).
The Ohio counties of Butler (267), Franklin (223), Hamilton (127) and Mongomery (202) have reported the most cases during this outbreak.
ODH took this action in response to the HepA outbreak reaching 1,657 confirmed cases, hospitalizations 1,042 (63%), and 5 deaths since January 2018.
Ohio represented approximately 15 percent of all HepA cases reported to the Centers for Disease Control and Prevention (CDC) during 2018 when 10,582 cases were confirmed in the USA.
The ODH press release in January 2019 recommends the following actions for Ohio residents:
- People who believe that they are at high risk for hepatitis A infection should contact their healthcare provider or local health department for information about vaccination
- People who know that they have been exposed to someone with hepatitis A should contact their healthcare provider or local health department to discuss post-exposure vaccination options
- Individuals who experience symptoms of hepatitis A should contact their healthcare provider
According to a June 2018 statement by the ODH, HepA is a vaccine-preventable, liver disease, often spread by contaminated food and water. And, ODH has provided more than 5,000 doses of hepatitis A vaccine to local health departments.
And, by declaring a hepatitis A outbreak ensures ODH access to additional vaccines through the CDC.
Recently, 2 additional risk profiles have been identified during the nationwide HepA outbreak: restaurant staff and homelessness.
During 2018, over 25 restaurants reported staff working while infectious with HepA.
And, the CDC formally adopted a recommendation that all persons aged 1 year and older experiencing homelessness should be routinely immunized against HepA.
In Ohio, doctors, nurses, and pharmacists can deliver the Hepatitis A vaccine to children 24 months and older and to adults.
To prevent the spreading of hepatitis, the CDC says there are 3 brands of hepatitis A vaccines available, Vaqta and Havrix. Both are approved for people older than 1 year of age, in a 2-dose series.
And, Twinrix is a combined hepatitis A and hepatitis B vaccine approved for people older than 18 years of age.
The CDC Vaccine Price List displays current hepatitis vaccine contract prices and general information. And, hepatitis vaccine discounts can be found here.
Vaccines, like any medicine, can have side effects. You are encouraged to report negative side effects of vaccines to the FDA or CDC.
Our Trust Standards: Medical Advisory Committee