Innovative Dengue Vaccine Candidate Granted Priority Review

Takeda announced yesterday that the U.S. Food and Drug Administration (FDA) had accepted and granted priority review for the tetravalent TAK-003 (QDENGA®) dengue vaccine candidate.
In the U.S., TAK-003 is being evaluated for the prevention of dengue disease caused by any dengue virus serotype in individuals four years through 60 years of age.
Recovery from infection by one serotype provides lifelong immunity against only that serotype. Conversely, later exposure to any remaining serotypes is associated with an increased risk of severe disease.
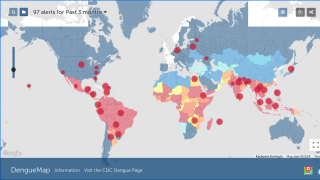
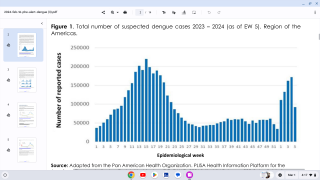
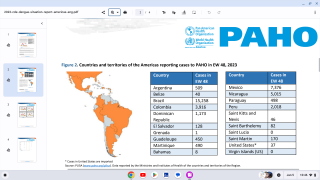
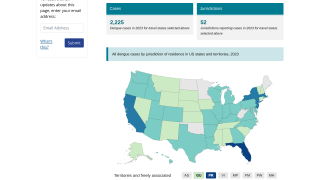
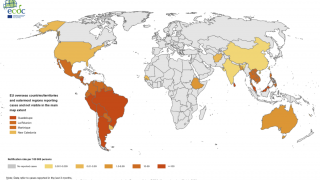
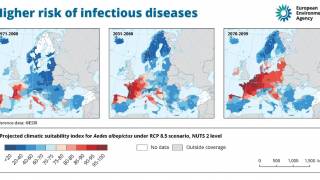
Dengue is a mosquito-borne virus endemic in more than 125 countries, including the U.S. territories of Puerto Rico, the U.S. Virgin Islands, and American Samoa.
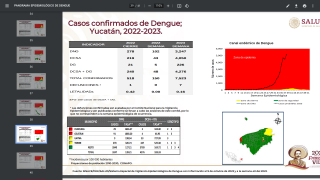
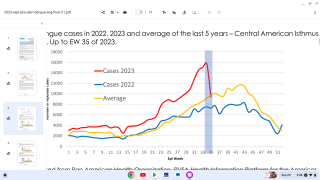
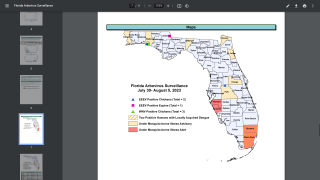
Incidence of locally-acquire dengue has increased in Florida during the summer of 2022. Aedes aegypti mosquitoes mainly spread dengue.
As of November 23, 2022, the only U.S. FDA-approved dengue vaccine is Dengvaxia.
Currently, TAK-003 has not been approved by the FDA or any other health authority outside of Indonesia. Other dengue vaccine news is posted at PrecisionVaccinations.com/Dengue.
Our Trust Standards: Medical Advisory Committee