Dengue Vaccines
Dengue Vaccines 2024
Dengue is a vaccine-preventable disease according to the World Health Organization (WHO), the U.S. Food and Drug Administration (FDA), the U.K. Medicines and Healthcare Products Regulatory Agency (MHRA), Pan American Health Organization (PAHO), the European Medicine Agency (EMA), Australia's Technical Advisory Group on Immunisation, and Brazil's National Health Surveillance Agency (ANVISA). These agencies recommend dengue vaccination for persons living or visiting dengue-risk areas. Since October 2022, two dengue vaccines have been authorized in various countries. Vaccine-efficacy studies for quadrivalent dengue-virus vaccines conducted in endemic countries have provided an excellent plan to guide the progress of phase 3 trials in India, contributing to about 30% of the global burden of dengue.
Dengvaxia® is a live attenuated tetravalent chimeric vaccine approved by the U.S. FDA and various countries. On June 30, 2023, the FDA issued a Supplemental Approval letter.
QDENGA® dengue vaccine is approved or authorized in various countries. In October 2023, the WHO recommended the QDENGA vaccine.
Dengue Vaccine Availability
As of April 2024, the second-generation dengue vaccine (QDENGA) was available in Argentina, Brazil, Denmark, Europe, Germany, India, Indonesia, Portugal, Thailand, and the U.K. Dengvaxia has been authorized in various countries since 2015.
Dengue Vaccine Candidates 2024
Butantan Institute Butantan-DV tetravalent dengue vaccine candidate showed an efficacy of 79.6% in preventing the disease in Brazil, according to a phase 3 clinical study, which continues through 2024. On February 1, 2024, an Original Article concluded a phase 3 clinical study found a single dose of Butantan-DV prevented symptomatic DENV-1 and DENV-2, regardless of dengue serostatus at baseline, through 2 years of follow-up. Butantan-DV is derived from a technology from the U.S. NIH licensed in 2009.
Serum Institute of India tetravalent dengue vaccine live candidate Dengusiil is conducting phase 1 clinical research in Australia. A study published in August 2023 reported that the vaccine was highly immunogenic. More than 69% of participants had tetravalent seroconversion, and more than 15 had trivalent seroconversion. SSI says Dengusiil will be commercialized in India, Pakistan, Bangladesh, Nepal, Bhutan, Maldives, and Sri Lanka.
National Institute of Allergy and Infectious Diseases - TetraVax-DV T005 (rDEN3Δ30/31-7164) is a live attenuated tetravalent vaccine. A phase 2 clinical trial revealed that, with three years of follow-up, a single dose of TV005 was well tolerated and immunogenic for all four serotypes in young children and adults, including individuals with no previous dengue exposure. Results of a phase 2 study of TV005 in Bangladesh published in 2024 reported the waning of antibody titres in children aged 1–4 years who were seronegative at the time of vaccination. Only 22–28% of children in this age group remained seropositive for DENV-1, DENV-3, and DENV-4 after three years of follow-up, compared with 69% seropositivity for DENV-2.
TetraVax-DV-TV003 (V180) is a live attenuated recombinant tetravalent investigational dengue vaccine conducting a phase 3 clinical trial in Brazil. Dr. Stephen Whitehead's laboratory developed the vaccine.
The DV1-DV4 vaccine candidate is transitioning into a human clinical study. This new vaccine construct, which comprises Nature's gene-chip peptides bound to a quantum cluster gold nanoparticle delivery system, was shown to have an excellent safety profile in a repeat dose Good Laboratory Practice grade toxicology study using a standard industry model.
Àvida Biotech's novel oral vaccine candidate for dengue does not require cold transport or storage and has completed proof of concept in a mouse model. In addition, the University of Buffalo's Center for Integrated Global Biomedical Sciences will provide preclinical drug development expertise.
K.M. Biologics KD-382 vaccine is a live attenuated tetravalent dengue candidate in phase 1 clinical t. It is expected to be effective against all four serotypes with a single dose. In addition, this live attenuated virus vaccine is expected to induce neutralizing antibodies and cellular immunity, similar to natural infection.
Emergex PepGNP-Dengue DengueTcP™, Its Novel T Cell-Priming Immune Set-Point Candidate, uses 100% synthetic vaccines to 'prime' naive CD8+ T-Cells to generate virus-specific CTL (CD8+ Cytotoxic T Lymphocyte cells) to kill infected cells before productive viral infection, thus preventing viral replication and disease in the vaccinated person. naNO-DENGUE: A Phase-I study of a nanoparticle-based peptide vaccine against Dengue virus.
CodaVax-DENV is a next-generation tetravalent live-attenuated dengue vaccine candidate under development by Codagenix Inc., whose vaccine design platform has enabled precise and rational attenuation of contemporary serotypes of all four strains of dengue virus through selective codon deoptimization. With this approach, Codagenix can rationally balance all four virus serotypes to produce a safe and highly immunogenic vaccine. On October 24, 2023, Codagenic announced that the U.S. Department of Defense awarded the Company $5.88 million to advance the development of its CodaVax-DENV program. The funding supports good manufacturing practices of drug substances and tetravalent drug products for a Phase 1 study and a first-in-human Phase 1 safety and immunogenicity trial. This award complements a $4.4 million DoD award granted in 2022.
Developed by Panacea Biotech in collaboration with the Indian Council of Medical Research, the Tetravalent Panacea dengue vaccine has launched phase l/ll trials in India. PTI reported a phase 3 trial is likely to begin in mid-2023.
Indian Immunologicals Limited (IIL) expects to launch its dengue fever vaccine commercially by 2026. IIL's managing director, K. Anand Kumar, said on August 20, 23, that the vaccine's early-stage trials conducted on about 90 individuals aged 18-50 did not demonstrate any adverse effects. The U.S. National Institute of Health provided IIL with the dengue virus required to develop the vaccine.
Dengue Vaccine Price
Pricing for Dengvaxia® is about $99 in the USA. In addition, the CDC's Vaccine for Children fund supports eligible people. And UNICEF offers various support programs worldwide. Additional vaccine price information is posted at InstantRx™. Market research forecasts an 11.7% CAGR through 2030, indicating that dengue vaccines will become over a $1.2 billion market segment.
Dengushield (VIS513) Monoclonal Antibody Therapy
Dengushield (VIS513) is a humanized monoclonal antibody (mAb) that delivers passive immunization and is being evaluated in phase 2 clinical trials for the treatment of dengue. VIS513 is a highly potent inhibitor of all four types of dengue viruses, both in vitro and in preclinical animal models. VIS513 was licensed to the Serum Institute of India Pvt. Ltd (SIIPL) for development and commercialization may cost 5,000 to 10,000 rupees per dose. SIIPL funded a phase 1 study announced in February 2024, and the VIS513 mAb was safe and well tolerated. It showed a dose-proportionate increase in pharmacokinetic exposure.
Dengue Antivirals
The Janssen Pharmaceutical Companies announced on October 20, 2023, promising data from a Phase 2a human challenge study evaluating JNJ-1802, a first-in-class oral antiviral in development to prevent dengue. The data showed that the compound induced antiviral activity against dengue (DENV-3) in humans, compared to placebo, and is safe and well-tolerated. This new study follows data published in the journal Nature in March 2023, which showed that JNJ-1802 provides strong protection against dengue in non-human primates and mice, and a Phase 1 first-in-human clinical study showing that the antiviral was safe and well-tolerated.
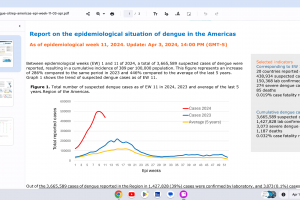
Dengue Outbreaks 2024
For the latest travel alerts regarding dengue outbreaks in 2024, visit Precision Vax.
Dengue Vaccine News
January 15, 2024 - The WHO has officially launched its 2024 Health Emergency Ap, seeking US$1.5 billion in funding.
December 15, 2023 - The journal PLOS Global Public Health published a study that found QDENGA had an efficacy of 61.2% against virologically confirmed dengue, 84% against hospitalizations, and 70% % against the development of dengue hemorrhagic fever at 54 months.
November 7, 2023 - The journal Nature published an article: Dengue is spreading. Can new vaccines and antivirals halt its rise?
June 22, 2023 - Wilbur Chen, MD, MSc, led the U.S. CDC's ACIP Workgroup presentations on dengue vaccines.
January 11, 2023 - The PAHO Technical Advisory Group on Immunization provided regional recommendations on vaccines against dengue.
Note: The content on this webpage is aggregated from the WHO, the U.S. CDC, research studies, and various media sources. Healthcare providers like Dr. Bob Carlso review this content for efficacy.