Three-Dose Malaria Vaccine Candidate Shows Promise
A recent article published by the Infectious Disease Special Edition highlights the safety and efficacy demonstrated by Sanaria's non-replicating whole parasite PfSPZ malaria vaccine candidate.
The new study involved 80 participants in a randomized controlled clinical trial comparing three doses of the PfSPZ vaccine against a placebo.
Vaccine efficacy was shown to be 48% at six months (P=0.061) and 46% at 18 months (P=0.018).
And two weeks after the last dose, antibodies to P. falciparum circumsporozoite protein and PfSPZ were higher in protected versus unprotected vaccinees.
Furthermore, a three-dose regimen of PfSPZ demonstrated safety and efficacy against malaria infection in malaria-experienced adults.
"Our study shows that the vaccine can be given to malaria-experienced adults in a highly endemic area and still provide protection, which is difficult and complicated as these individuals already have significant immune responses to malaria parasites that a vaccine must overcome for it to be successful," wrote the University of Maryland School of Medicine's Center for Vaccine Development and Global Health.
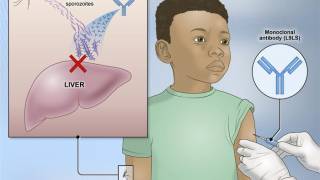
Sanaria Inc.'s PfSPZ vaccine candidate is made with a live-attenuated form of the malaria parasite Plasmodium falciparum sporozoite, which is transmitted by mosquitos.
A previous study with African adults found PfSPZ provided 52% vaccine efficacy against naturally transmitted malaria infection, and protection lasted 8 to 14 months.
"New strategies are needed to achieve the United Nations sustainable development goal of a 90% reduction in malaria incidence and mortality by 2030," commented Mark T. Gladwin, MD, Vice President for Medical Affairs, University of Maryland, Baltimore, in a press release on December 22, 2022.
"Innovative vaccines that provide a higher level of protection against malaria are urgently needed to help to achieve this goal."
According to the World Health Organization, about 240 million people were infected with malaria in 2020, and 627,000 people died from their infections,
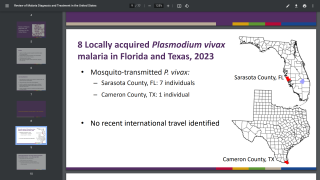
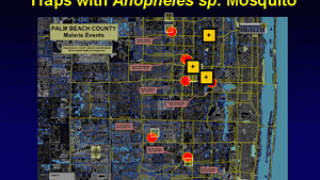
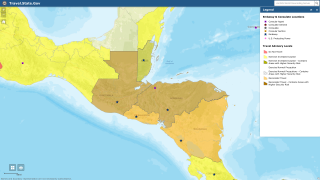
The vast majority of malaria cases in the U.S. are in travelers returning from countries where malaria transmission occurs, such as sub-Saharan Africa and South Asia, says the U.S. Centers for Disease Control; and Prevention (CDC).
About 2,000 cases of malaria are diagnosed in the U.S. each year.
The CDC says malaria is a vaccine-preventable mosquito-borne disease caused by a parasite.
In October 2021, the World Health Organization recommended the Mosquirix™ (RTS,S) malaria vaccine for regions with moderate to high malaria transmission.
And in September 2022, India granted permission to send two lakh doses of the R21/Matrix-M™ malaria vaccine to the U.K.
However, as of January 9, 2023, the U.S. Food and Drug Administration has not approved either malaria vaccine currently in use.
The FDA did approve Artesunate for Injection™ to treat severe malaria in May 2020.
Disclosures: The researchers did not disclose any industry conflicts.
Our Trust Standards: Medical Advisory Committee