A Different Type of RSV Vaccine

A clinical-stage synthetic biology company with a rational virus design platform for viral vaccines and immuno-oncology therapeutics today announced that it has initiated dosing in a pediatric Phase 1 study evaluating CodaVax™-RSV, a live-attenuated, intranasal vaccine candidate for the prevention of disease caused by the Respiratory Syncytial Virus (RSV).
In contrast with inactivated RSV vaccines, live RSV vaccine viruses do not appear to prime for enhanced RSV disease based on animal and natural history studies.
As such, live-attenuated vaccines appear to be the type of RSV vaccines that have been demonstrated to be safe in RSV-naïve recipients.
"Codagenix is focused on providing an effective prophylactic vaccine for infants and toddlers six months to 5 years old who, based on the recent data, may be at higher risk for hospitalization following infection despite recent innovations in other RSV vaccines for the elderly that are under review," said J. Robert Coleman, Ph.D., Co-founder and Chief Executive Officer of Codagenix, in a press release on May 10, 2023.
"As a live-attenuated, intranasal vaccine designed using our differentiated codon deoptimization platform, CodaVax™-RSV is particularly well positioned to address this significant unmet need since it has the potential to induce innate immunity and durable local and systemic immunity."
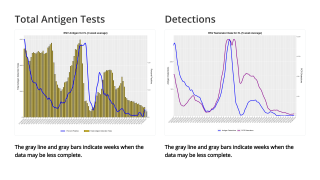
While successful Phase 3 efficacy trials have been announced for the population over 60, a heavier burden of disease, as measured by hospitalization in the 2022-23 RSV season, was in the 6-month to 1-year-old segment.
Specifically, the >65 aged population saw a peak RSV-associated hospitalization of 6.5 per 100,000 compared to the 6-month to the 1-year-old population that experienced an RSV-associated hospitalization rate of 99.1 per 100,00, representing a 15-fold higher burden in the pediatric population.
Further, maternal immunization and long-acting antibody strategies have shown potential effectiveness in protecting those < 6 months of age, still leaving an unmet need for protecting older infants and toddlers in the 6-month to 5-year-old age bracket.
This study is a Phase 1, randomized, double-blind, placebo-controlled, dose-escalation clinical trial to evaluate the safety of and immune response to CodaVax-RSV in healthy children.
They will be vaccinated in spring to early autumn 2023 and followed through the 2023-24 RSV season.
Children will receive 2 doses of the vaccine at one of several dose levels or placebo (saline solution with no active ingredient) as nose drops; doses will be 28 days apart.
And a safety committee will review the safety profile of each dosing group before the next dose escalation.
According to the U.S. Centers for Disease Control and Prevention, it is estimated that 58,000 to 80,000 children aged five and younger are hospitalized each year due to an RSV infection.
This data indicates a more significant disease burden than influenza.
RSV vaccine news is posted by Precision Vaccinations.
Our Trust Standards: Medical Advisory Committee