Dengue Vaccine Dengvaxia Approved in the USA

Dengvaxia became the first vaccine approved for the prevention of dengue disease in people ages 9 through 16, who have laboratory-confirmed previous dengue infection, and who live in endemic areas caused by all dengue virus serotypes.
Sanofi Pasteur’s Dengvaxia is a live, attenuated vaccine that is administered as 3 separate injections, with the initial dose followed by two additional shots given six and twelve months later.
This U.S. Food and Drug Administration (FDA) announcement on May 1, 2019, is important since there is no cure for dengue disease.
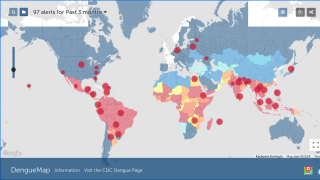
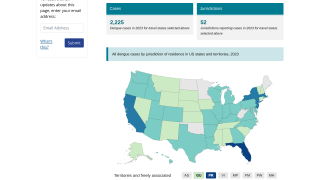
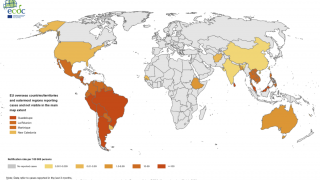
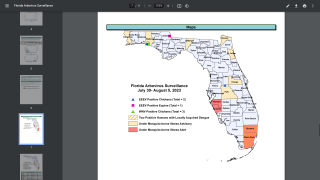
Although dengue cases are rare in the continental USA, the disease is regularly found in American Samoa, Puerto Rico, Guam, the U.S. Virgin Islands, as well as Latin America, Southeast Asia, and the Pacific Islands.
“Dengue disease is the most common mosquito-borne viral disease in the world and global incidence has increased in recent decades,” said Anna Abram, FDA deputy commissioner for policy, legislation, and international affairs.
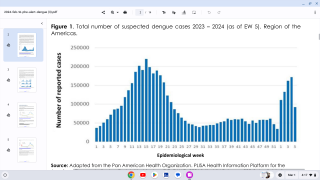
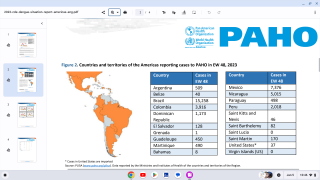
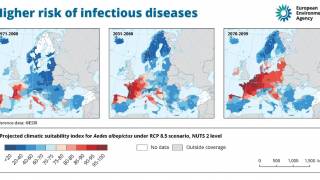
The U.S. Centers for Disease Control and Prevention (CDC) estimates more than one-third of the world’s population is living in areas at risk for infection by dengue virus which causes dengue fever, a leading cause of illness among people living in the tropics and subtropics.
The safety and effectiveness of the Dengvaxia vaccine were determined in 3 randomized, placebo-controlled studies involving approximately 35,000 individuals in dengue-endemic areas, including Puerto Rico, Latin America, and the Asia Pacific region.
The vaccine was determined to be approximately 76 percent effective in preventing symptomatic, laboratory-confirmed dengue disease in individuals 9 through 16 years of age who previously had laboratory-confirmed dengue disease.
Dengvaxia has already been approved in 19 countries and the European Union.
The most commonly reported side effects by those who received Dengvaxia were a headache, muscle pain, joint pain, fatigue, injection site pain, and low-grade fever.
Dengvaxia is not approved for use in individuals not previously infected by any dengue virus serotype or for whom this information is unknown.
This is because, in people who have not been infected with dengue virus, Dengvaxia appears to act as a first dengue infection – without actually infecting the person with wild-type dengue virus – such that a subsequent infection can result in severe dengue disease.
Therefore, healthcare professionals should evaluate individuals for prior dengue infection to avoid vaccinating individuals who have not been previously infected by the dengue virus.
This can be assessed through a medical record of a previous laboratory-confirmed dengue infection or through serological testing (tests using blood samples from the patient) prior to vaccination.
Currently, no available tests have been cleared by the Food and Drug Administration (FDA) to determine a previous dengue infection.
Which means, you should speak with your healthcare professional about available dengue tests.
Related dengue virus news:
- Guam Joins Tahiti and Fiji Reporting Dengue Fever Cases in 2019
- Dengue Outbreak in the Philippines Exceeds 52,000 Cases During 2019
The FDA, an agency within the U.S. Department of Health and Human Services, protects the public health by assuring the safety, effectiveness, and security of human and veterinary drugs, vaccines and other biological products for human use, and medical devices.
Relevant Links: CDC vaccination schedules, CDC price list, how to report vaccine side effects, and international travel alerts.
Our Trust Standards: Medical Advisory Committee