Ebola Vaccine Now Available in African Countries

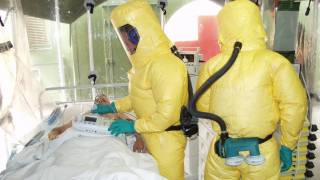
Four African countries announced their approval of the Zaire Ebolavirus vaccine named ERVEBO.
The ERVEBO vaccine is now been registered by National Health Authorities in the Democratic Republic of Africa (DRC), Burundi, Ghana, and Zambia.
These approvals announced on February 14, 2020, signify continued, groundbreaking progress in advancing the future of global public health preparedness against Zaire ebolavirus disease.
And, were the result of the successful implementation of the World Health Organization (WHO) Roadmap for introduction and roll‐out of Merck rVSV-ZEBOV (v920) Ebola virus disease vaccine candidate in African countries.
The roadmap, designed to coordinate actions and contributions toward the licensing and roll-out of ERVEBO, helped facilitate near-parallel regulatory reviews and led to the approvals of the vaccine in several at-risk countries within 90 days of WHO prequalification.
According to Merck, the producer of ERVBO, this progress is made possible by the unprecedented collaboration between the WHO, the African Vaccines Regulatory Forum, African governments, the European Medicines Agency (EMA), and Merck.
“We are grateful for WHO’s leadership in establishing a path forward for expediting the prequalification and licensing of this vaccine in countries at greatest risk,” said Kenneth C. Frazier, chairman, and chief executive officer, Merck, in a press release.
“This important milestone is one more example of the partnership that has formed in response to the Ebola outbreaks.”
“While we are far from finished in the Ebola fight, this milestone shows what can be done when we work together to address the most challenging diseases that threaten people and communities.”
As previously announced, Merck is working to initiate the manufacturing of licensed doses and expects these doses to start becoming available in approximately the third quarter of 2020.
Merck is working closely with the United States government, WHO, UNICEF, and Gavi to plan for how eventual, licensed doses will support future public health preparedness and response efforts against Zaire ebolavirus disease.
In the meantime, Merck continues to work urgently with WHO and partners to make investigational Ebola Zaire vaccine doses available in support of international outbreak response efforts in the DRC and neighboring countries.
ERVEBO was granted conditional marketing authorization by the European Commission on November 11, 2019, and approved by the U.S. Food and Drug Administration (FDA) on Dec. 20, 2019.
The FDA says ERVEBO is indicated for the prevention of disease caused by Zaire ebolavirus in individuals 18 years of age and older. The duration of protection conferred by ERVEBO is unknown.
Additionally, the effectiveness of the ERVEBO vaccine is unknown when administered concurrently with antiviral medication, immune globulin, and/or blood or plasma transfusions says the FDA.
And, the ERVEBO vaccine does not protect against other species of Ebolavirus or Marburgvirus.
Recently, a new Health Advisory was updated with guidance regarding the usage of a new rapid diagnostic test (RDT) for detecting the Ebola virus in symptomatic patients.
Health Advisory (#432) says ‘healthcare providers caring for a patient with possible Ebola virus infection should first contact their local or state public health authorities before any testing is performed.’
‘RDTs should be used only in cases where more sensitive molecular testing is not available.’
And, ‘the interpretation of RDT results should be done with caution and in consultation with relevant public health authorities to ensure appropriate testing and interpretation of results.’
This advisory says ‘there have been 49 travelers since August 2018 who were ill when returning to the USA from the DRC or the surrounding African countries.
Of these 49 individuals, testing for Ebola was recommended for just 1 person, who was found not infected.
Ebola vaccine and treatment news published by Precision Vaccinations.
Our Trust Standards: Medical Advisory Committee