Ebola Vaccine Combo Gains Positive Opinion

The European Medicines Agency (EMA) announced a positive opinion has been issued for an innovative Ebola vaccine regimen for the prevention of the Ebola Virus Disease caused by the Zaire ebolavirus species.
Marketing Authorisation Applications (MAAs) were submitted to the EMA in support of the vaccines in the 2-dose regimen (Zabdeno (Ad26.ZEBOV), Mvabea (MVA-BN-Filo).
The goal of this two-dose vaccination approach is to induce long-term immunity against Ebola Virus Disease (EVD).
Johnson & Johnson and its Janssen Pharmaceutical Companies said they received on May 29, 2020, a positive opinion from the Committee for Medicinal Products for Human Use (CHMP).
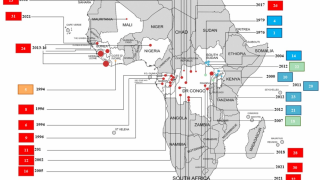
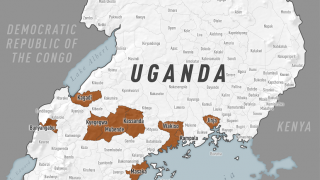
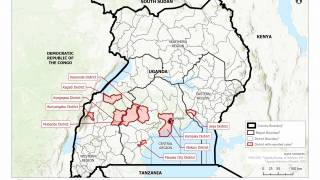
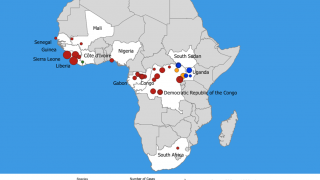
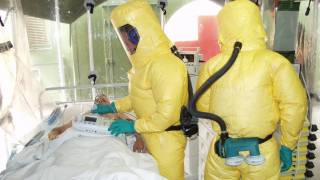
This is important news since the most recent Ebola outbreak, which started in the Democratic Republic of the Congo (DRC) in central Africa during August 2018, has reported a mortality rate of 65 percent.
Janssen’s investigational Ebola vaccine regimen is specifically designed to support preventive vaccination in countries that are at risk of Ebola outbreaks, as well as for other at-risk groups such as healthcare workers.
To date, approximately 60,000 people have been vaccinated with Janssen’s investigational preventive Ebola vaccine regimen in clinical studies and vaccination initiatives.
Janssen-sponsored Phase 1 studies have been reported in peer-reviewed journals including JAMA and the Journal of Infectious Diseases, and Phase 1, 2 and 3 data were recently presented at the 2019 European Congress of Clinical Microbiology & Infectious Disease (ECCMID).
These studies indicate that the vaccine regimen is well-tolerated, inducing robust and durable immune responses to the Zaire ebolavirus strain.
In May 2019, the WHO’s Strategic Advisory Group of Experts (SAGE) on immunization recommended the use of the Janssen investigational Ebola vaccine regimen as part of efforts to contain the DRC outbreak10 and more than 50,000 people in the DRC11 and Rwanda12 were vaccinated.
The regimen includes Ad26.ZEBOV as the first dose, based on Janssen’s proprietary AdVac® viral vector technology, and MVA-BN-Filo as the second dose, based on Bavarian Nordic’s MVA-BN® technology, administered approximately 8-weeks later.
The Janssen investigational preventive Ebola vaccine regimen (Ad26.ZEBOV, MVA-BN-Filo) utilizes a viral vector strategy in which viruses – in this case adenovirus serotype 26 (Ad26) and Modified Vaccinia Virus Ankara (MVA) – are genetically modified so that they cannot replicate in human cells.
In addition, these vectors are modified to safely carry the genetic code of an Ebola virus protein in order to trigger an immune response.
Johan Van Hoof, M.D., Global Therapeutic Area Head, Vaccines, and Managing Director, Janssen Pharmaceutica N.V., said in a related press statement, “If our investigational Ebola vaccine regimen is approved by the European Commission, this would be Janssen’s first vaccine approval and an important step forward in our efforts to help protect people at risk of Ebola Virus Disease.”
J&J says ‘discussions with the FDA are ongoing to define the required data set for filing Janssen’s Ebola vaccine regimen under the FDA’s Animal Rule licensure pathway.’
Janssen’s investigational vaccine regimen originates from a collaborative research program with the NIH and received direct funding and preclinical services from the National Institute of Allergy and Infectious Diseases, part of NIH, under Contract Number HHSN272200800056C.
Further funding for the Ebola vaccine regimen has been provided in part with federal funds from the Office of the Assistant Secretary for Preparedness and Response, BARDA under Contract Numbers HHSO100201700013C and HHSO100201500008C.
At Janssen, the Pharmaceutical Companies of Johnson & Johnson, working tirelessly to make that future a reality for patients everywhere by fighting sickness with science, improving access with ingenuity, and healing hopelessness with heart.
Ebola vaccine news published by Precision Vaccinations.
Our Trust Standards: Medical Advisory Committee