Hexavalent Vaccine To Be Added to the Vaccines for Children Program

The US government’s vaccine advisory committee voted unanimously to include all parts of a hexavalent vaccine VAXELIS within the Vaccines for Children (VFC) program.
This is good news since combination vaccinations have been associated with increased coverage and higher completion rates in children, says the Centers for Disease Control and Prevention (CDC).
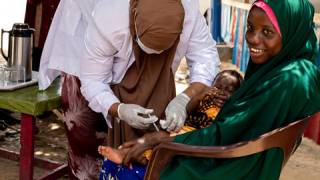
The VAXELIS vaccine is a 3-dose series for children prior to their 5th birthday and is indicated for active immunization to prevent diphtheria, tetanus, pertussis, poliomyelitis, hepatitis B, and invasive disease due to Haemophilus influenzae type b.
VAXELIS was developed jointly by Sanofi and Merck and has been approved by the Food and Drug Administration for use in children through age 4 years of age on December 21, 2018.
This announcement by the Advisory Committee on Immunization Practices (ACIP) means the VAXELIS vaccine will be included in the infant series at ages 2, 4 and 6 months, at no cost to children whose families cannot afford vaccination, reported Katherine Bortz, with Healio.
Unfortunately, the VAXELIS vaccine may not become available until 2021.
The Vaccines for Children Program helps ensure that all children have a better chance of getting their recommended vaccinations that protect them from 16 diseases.
A child is eligible for the VFC Program if he or she is younger than 19 years of age and is one of the following: Medicaid-eligible, Uninsured, Underinsured, and American Indian or Alaska Native.
The funding for the VFC program is approved by the Office of Management and Budget and allocated through the Centers for Medicare & Medicaid Services to the CDC.
The CDC buys vaccines at a discount and distributes them to state health departments and certain local and territorial public health agencies—which in turn distribute them at no charge to those private physicians’ offices and public health clinics registered as VFC providers.
This June 27, 2019, ACIP vote will be officially announced in an MMWR policy note published later in 2019.
Our Trust Standards: Medical Advisory Committee