Takeda’s Dengue Vaccine Met Phase 3 Goals

The pivotal Phase 3 clinical trial of dengue vaccine candidate TAK-003 was reported to have met the primary efficacy endpoint.
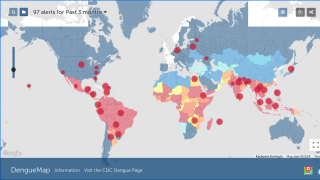
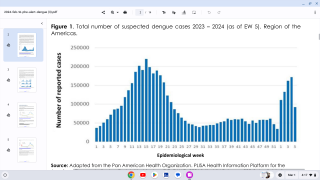
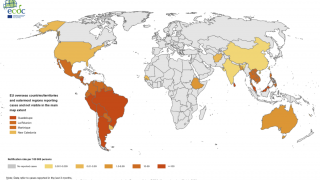
This is important news since approximately half of the world’s population lives under the threat of the dengue fever virus, which is estimated to cause 400 million infections and 20,000 deaths globally each year, says the World Health Organization (WHO).
Currently, the TAK-003 vaccine is not licensed by any country.
This first analysis of the Tetravalent Immunization against Dengue Efficacy Study (TIDES) trial showed that Takeda’s investigational live-attenuated tetravalent dengue vaccine TAK-003 was efficacious in preventing dengue fever caused by any of the 4 serotypes of the virus.
The TIDES trial, Takeda’s largest interventional clinical trial to date, enrolled over 20,000 healthy children and adolescents ages four to 16 years living in dengue-endemic areas.
This Phase 3 study was designed to evaluate the efficacy, safety and immunogenicity of 2 doses of TAK-003, in both dengue exposed and naïve individuals.
The TIDES trial is continuing and additional results are expected later this year, along with results from other Phase 3 studies.
Rajeev Venkayya, M.D., President of the Global Vaccine Business Unit at Takeda, said in a press release, “We are excited to publish the data in a peer-reviewed journal as quickly as possible. In parallel, we are advancing the clinical development, commercial manufacturing, and stakeholder consultations to support a potential future global launch of the vaccine.”
Previous Phase 1 and 2 study data showed that TAK-003 induced immune responses against all 4 dengue serotypes, in both seropositive and seronegative participants, and the vaccine was found to be generally safe and well tolerated.
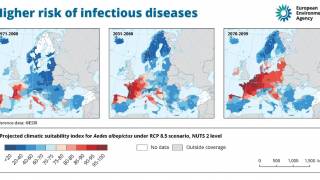
Dengue is a mosquito-borne viral disease that is transmitted by female mosquitoes mainly of the species Aedes aegypti and, to a lesser extent, Ae. albopictus.
Dengue is widespread throughout the tropics, with local variations in risk influenced by rainfall, temperature and unplanned rapid urbanization.
Severe dengue, also known as Dengue Haemorrhagic Fever, was first recognized in the 1950s during dengue epidemics in the Philippines and Thailand.
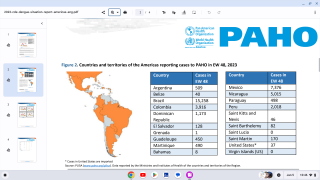
Today, severe dengue affects most Asian and Latin American countries and has become a leading cause of hospitalization and death among children and adults in these regions.
There are 4 distinct but closely related, serotypes of the virus that cause dengue (DEN-1, DEN-2, DEN-3, and DEN-4), says the WHO.
Recovery from infection by one provides lifelong immunity against that particular serotype.
However, cross-immunity to the other serotypes after recovery is only partial and temporary. Subsequent infections by other serotypes increase the risk of developing severe dengue.
Recent dengue virus news
- Dengue Fever Virus Alert Reaches Dominica Island
- Prior Dengue Infection May Protect Children From the Zika Virus
- Dengvaxia Vaccine Remains Banned in the Philippines
Another dengue vaccine, DengVaxia, was granted marketing authorization by the European Commission during December 2018.
And, DengVaxia is currently under priority review by the US Food and Drug Administration.
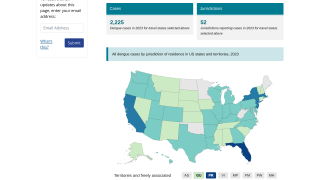
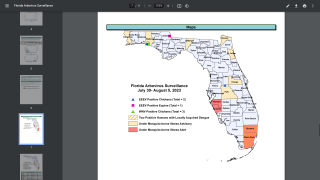
In the USA, Florida has been reporting local and travel-related dengue virus cases during 2018.
Americans planning international trips to dengue-endemic areas can schedule a vaccination counseling session with a local pharmacy at Vax-Before-Travel.
Our Trust Standards: Medical Advisory Committee