Mpox Outbreaks Have Not Ended
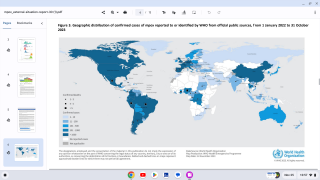
While the international community has focused on the extensive mpox outbreak in the Democratic Republic of the Congo, this sexually transmitted disease has resurfaced in the United States.
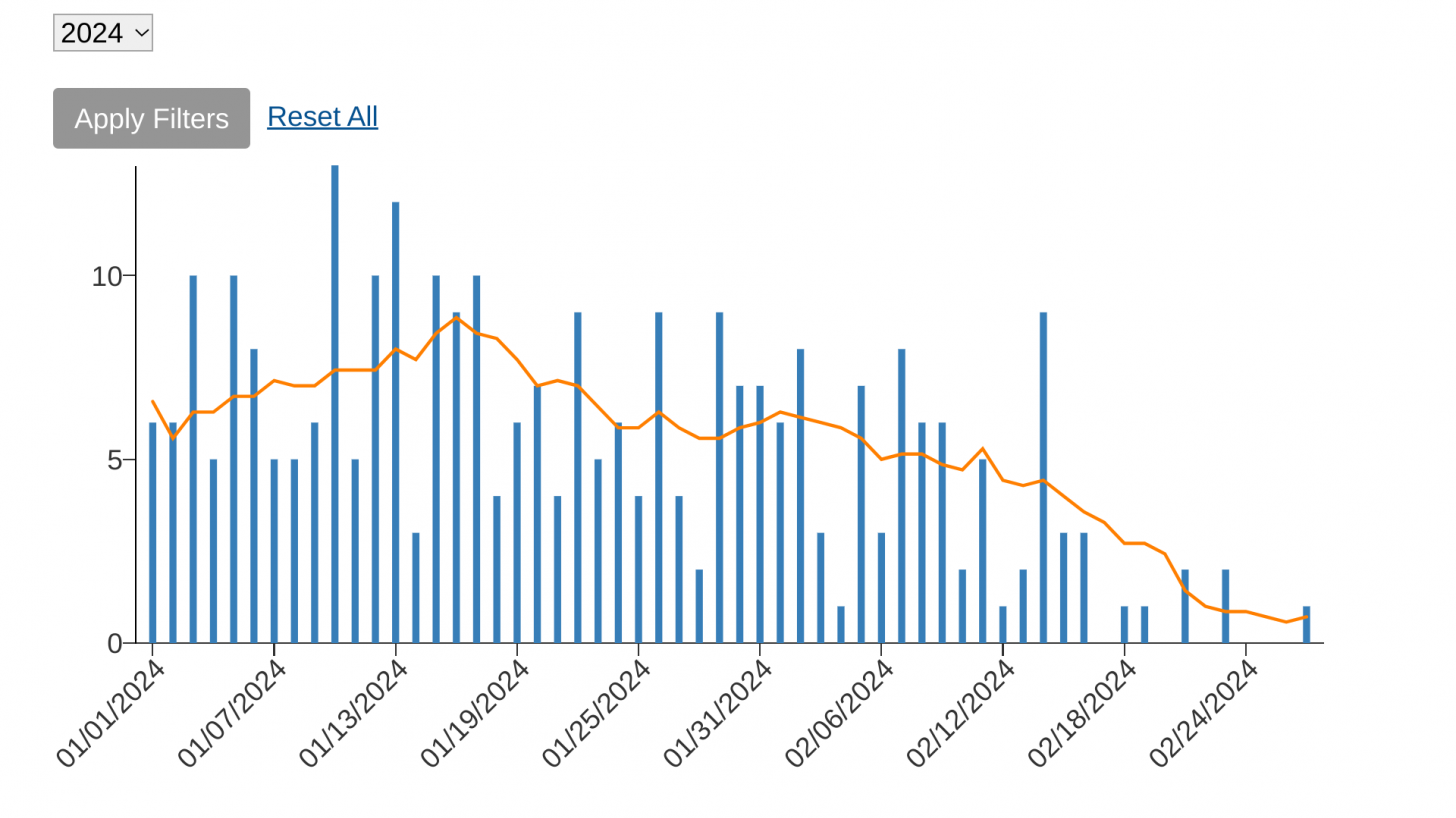
According to the U.S. Centers for Disease Control and Prevention (CDC), as of the week ending March 30, 2024 (Week #13), there were 664 mpox cases in the U.S.
New York (142), California (67), and Texas (43) are the leading states reporting mpox cases in 2024.
This data compares with just 307 reported in the same period in 2023.
To increase detections, LabCorp announced on April 10, 2024, that the U.S. FDA granted Emergency Use Authorization for its Mpox PCR Test Home Collection Kit to aid in the diagnosis of infection with non-variola Orthopoxvirus, including the mpox virus.
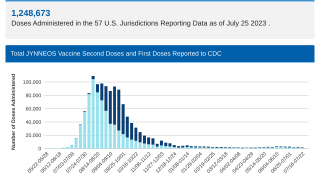
From a prevention perspective, Bavarian Nordic, manufacturer of the U.S. FDA-approved JYNNEOS® (MVA-BN®, IMVAMUNE®) vaccine, recently increased access by entering the commercial market.
As of April 1, 2024, JYNNEOS became more available in clinics and retail pharmacies in the U.S. This indicates pharmacists located in mpox hot spots will be able to meet the needs of more patients in 2024.
"We have done a significant amount of work to expand access to the vaccine in places where people want to be vaccinated. We know that pharmacy is one of the most important channels for people, so we focused on making it available there," Lee Ann Kimak, vice president, North America and head of U.S. Commercial for Bavarian Nordic, informed Precision Vaccination News on April 10, 2024.
"We have contracted with major pharmacies in the U.S., including Walgreens, which has hundreds of HIV Centers of Excellence."
"Also, we are focused on availability through other large retail chains, grocery, and big box stores."
Seperately, the U.S. CDC's Emerging Infectious Diseases Volume 30, Number 5 May 2024 edition, revealed on April 10, 2024, that, among vaccine-eligible GBMSM in the United Kingdom, mpox vaccine uptake was 69% (95% CI 65%–72%; 601/875) and 92% (95% CI 89%–94%; 601/655) among those offered the JYNNEOS vaccine.
The CDC says vaccination is an essential tool in preventing the spread of mpox and protects against severe illness, hospitalization, and death.
The current mpox outbreak in the DRC differs from the U.S. primarily because of a different mpox strain.
The Clade I strain is typically more severe, with about 7% of patients dying from the disease.
Unfortunately, about 2 out of 3 cases in the DRC are children under 15 years of age.
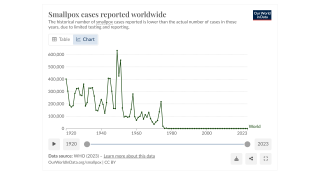
Since January 2023, DRC has reported 18,922 suspected mpox cases and 1,007 deaths.
The CDC says no Clade 1 cases have been reported in the United States at this time. Therefore, the risk to the general public in the U.S. from the type of mpox is low.
Furthermore, people who have already had mpox or are fully vaccinated with two doses of JYNNEOS should be protected against mpox Clade 1. Casual contact, such as that that might occur while traveling in Africa, is not likely to cause the disease to spread.
The best protection against mpox is two doses of the JYNNEOS vaccine, says the CDC.
Our Trust Standards: Medical Advisory Committee