$40 Million Funds New Generation of Biologics Targeting Virus Infections and Rabies

YS Biopharma Co., Ltd. today announced that it has entered into a share purchase agreement with an institutional investor for a private placement for an aggregate of US$40 million in proceeds.
As of February 9, 2024, YS Biopharma has developed a proprietary PIKA® immunomodulating technology platform and a new generation of preventive and therapeutic biologics targeting Rabies and other virus infections.
According to the World Health Organization, Rabies is a vaccine-preventable viral disease found in more than 150 countries and territories.
The PIKA rabies vaccine candidate is a lyophilized human-use rabies vaccine composed of cell culture-derived rabies antigen mixed with PIKA adjuvant, which acts as a TLR3 agonist. It is designed to induce accelerated and strong cellular immunity and rapidly stimulate the body to produce a higher humoral immune response.
And its accelerated onset of immune response allows a three-visit, one-week regimen superior to the currently available vaccine with a five-visit, one-month or three-visit, three-week regimen, which shortens the treatment period by two to three weeks.
The clinical studies to date have shown that PIKA rabies vaccine can achieve protective level of neutralizing antibodies as early as seven days post vaccination and elicit more robust immunogenic response compared to that of the control arm vaccine, which is a widely used commercially available vaccine.
On October 31, 2023, the company completed enrollment in its Phase 3 clinical trial, which will assess the safety, immunogenicity, and lot-to-lot consistency of the PIKA Rabies Vaccine.
On June 1, 2023, the Food and Drug Administration of the Philippines granted Phase 3 clinical trial approval, and on May 16, 2023, Pakistan issued study approval.
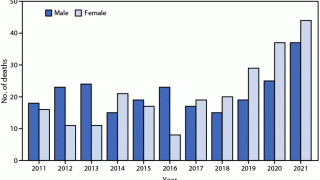
In the U.S., dog control programs were initiated in the 1940s. Since then, routine rabies vaccination eliminated the canine rabies virus variant from circulation by 2008.
As of 2024, bats are the leading cause of rabies deaths in people in all 49 continental U.S. states. Currently, several rabies vaccines for people, such as Chirorab®, are available worldwide.
Our Trust Standards: Medical Advisory Committee