Chikungunya Vaccine Candidate Approaches Authorization

France-based Valneva SE announced that it had completed recruitment for the clinical lot-to-lot consistency Phase 3 trial of its single-shot chikungunya vaccine candidate, VLA1553.
VLA1553 is the only chikungunya vaccine candidate conducting Phase 3 clinical trials as of June 10, 2021.
The lot-to-lot consistency Phase 3 trial runs parallel to the ongoing, pivotal Phase 3 trial, VLA1553-301, for which the Company already announced recruitment completion in April 2021.
Juan Carlos Jaramillo, Chief Medical Officer of Valneva, commented in a press release, “We are pleased to have reached this new recruitment milestone. We’ve now enrolled all participants for both our pivotal Phase 3 trial and lot-to-lot consistency trial so our VLA1553 program is progressing extremely well. Chikungunya virus is a major, growing public health threat, and we are looking forward to our top-line data this summer”.
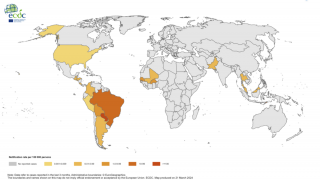
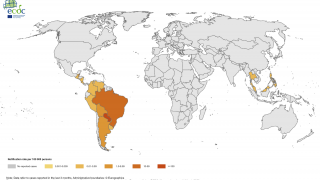
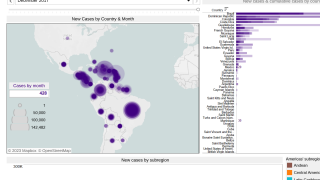
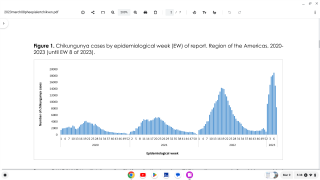
Chikungunya is a mosquito-borne viral disease caused by the chikungunya virus (CHIKV), a Togaviridae virus, transmitted by Aedes mosquitoes. An infection leads to symptomatic disease in 72-92% of humans after 4 to 7 days following the mosquito bite. As of September 2020, there were more than 3 million reported cases in the Americas.
There are no preventive vaccines or effective treatments available and, as such, chikungunya is considered to be a major public health threat.
The Company has also received confirmation for its proposal to seek licensure under the accelerated approval pathway from the FDA. Under this pathway, Valneva plans to seek licensure of the vaccine based on a surrogate of protection agreed with the FDA that is reasonably likely to predict protection from chikungunya infection.
The program was granted Fast Track designation by the FDA in December 2018 and PRIME designation by the European Medicines Agency in October 2020.
Valneva is a specialty vaccine company focused on developing and commercialization of prophylactic vaccines for infectious diseases with significant unmet medical needs.
Our Trust Standards: Medical Advisory Committee