Maternal Zika Viremia: a Marker of Adverse Perinatal Outcomes

The recent worldwide Zika virus (ZIKV) outbreak confirmed maternal-fetal transmission and its association with adverse perinatal outcomes.
Whether prolonged viremia after ZIKV infection in pregnant women represents a risk factor for maternal-fetal transmission, congenital Zika syndrome, or other adverse outcomes is an ongoing controversy.
The main findings of a recent ZIKV infection cohort study published by the US Centers for Disease Control and Prevention (CDC) on February 23, 2021, are 2-fold.
First, maternal ZIKV infection with prolonged viremia is associated with a 7-fold increased risk for fetal or neonatal adverse outcomes compared with pregnancies without prolonged viremia.
Second, prolonged maternal viremia is associated with a 2-fold increased risk for confirmed congenital infection than infected mothers without prolonged viremia.
The infected group’s sample size limited this study because only patients with a positive ZIKV RT-PCR result at enrollment were included in the analysis and followed up with monthly RT-PCR testing until clearance or delivery.
However, because the measured effect size was high, the sample size was sufficient to identify an association.
However, the limited number of prolonged viremia cases forced these researchers to group all adverse outcomes to analyze with sufficient power. It prevented the evaluation of its association with individual signs or symptoms or new characteristics.
Although ZIKV testing was based on the previous guidelines relevant during the 2015–2016 epidemic with adaptation to local capacities, testing does not follow the more recent CDC guidelines (patients considered as noninfected underwent serologic testing in each trimester rather than nucleic acid testing, as is currently recommended).
However, patients included as noninfected remained negative for IgM and IgG in each trimester and at delivery, limiting the risk for exposure misclassification.
Similarly, women with only positive IgM testing were excluded from the study because some might have had undetected prolonged viremia, which would have led to exposure misclassification and an underestimation of the consequences of prolonged viremia.
Information about the sensitivity and specificity of neonatal testing remains limited, and several studies have shown the progressive disappearance of ZIKV RNA in the maternal-fetal compartments.
Although the identification of IgM in fetal or neonatal blood was used to avoid congenital ZIKV false negatives, these researchers cannot exclude an outcome misclassification because ZIKV could have infected some neonates with negative results without viremia and immunity against ZIKV detectable at birth.
This risk is likely low given that >80% of fetuses or neonates from infected pregnant women underwent testing in >3 different samples (including blood, urine, placenta, cerebrospinal, and amniotic fluid) (19), and all neonates from non-infected pregnant women underwent serologic testing at birth.
Undetected congenital infections in the two reference groups might result in overestimation of the effect of prolonged viremia overallNeonates from noninfected mothers who underwent postnatal transfontanellar ultrasound (as well as by computed tomography and magnetic resonance imaging, if available) only in the case of an abnormal prenatal ultrasound or symptoms at birth, in contrast to those from infected mothers who underwent routine postnatal imaging.
Moreover, when they are asymptomatic, some neonates from noninfected mothers might have undetected cerebral anomalies at birth, resulting in an overestimation of the consequences of prolonged Zika viremia.
All neonates from noninfected mothers underwent multiple prenatal ultrasound assessments (enhanced by 2 supplementary examinations with neurosonograms during the epidemic), reducing the risk for undetected cerebral anomalies.
Negative and positive predictive values of prolonged maternal viremia for congenital infections and adverse outcomes related to ZIKV seem to be moderate because fetal and neonatal adverse outcomes and congenital infections also occur in pregnant women without identified prolonged viremia.
One explanation could be that prolonged viremia might reflect viral replication in the placenta without further involvement for the fetus.
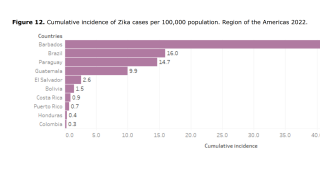
Some of our cases with prolonged maternal viremia (6/15) did not exhibit congenital infections, suggesting that prolonged maternal viremia might also reflect persistent viral replication in other reservoirs than the fetus or the placenta.
The study of Rodo et al. and a CDC report also described fetuses without congenital infection or adverse outcomes from mothers with prolonged viremia.
These researchers stated, ‘Our results also indicate that noninfected women exhibited a 5.1% risk for fetal neurologic anomalies and 1.2% risk for fetal loss, reflecting that other etiologies for adverse perinatal outcomes remain present even in the context of a ZIKV epidemic.
In conclusion, prolonged maternal ZIKV viremia could be a marker for an increased risk for maternal–fetal transmission and subsequent adverse perinatal outcomes.
Even if prolonged maternal viremia is not consistently present in congenital infection, it might reflect active viral replication in the fetal–placental compartment. It should lead to an enhanced prenatal and neonatal follow-up.
Acknowledgments: This study involved personnel at Centre Hospitalier de l’Ouest Guyanais and Institut Pasteur of French Guiana. The corresponding author attests that all listed authors meet authorship criteria. No others meeting the criteria have been omitted. This manuscript is an honest, accurate, and transparent account of the study being reported, and no important aspects of the study have been omitted.
ZikaNews publishes research-based Zika virus news.
Our Trust Standards: Medical Advisory Committee