Saliva-Based COVID-19 Test Returns Results in 45 Minutes
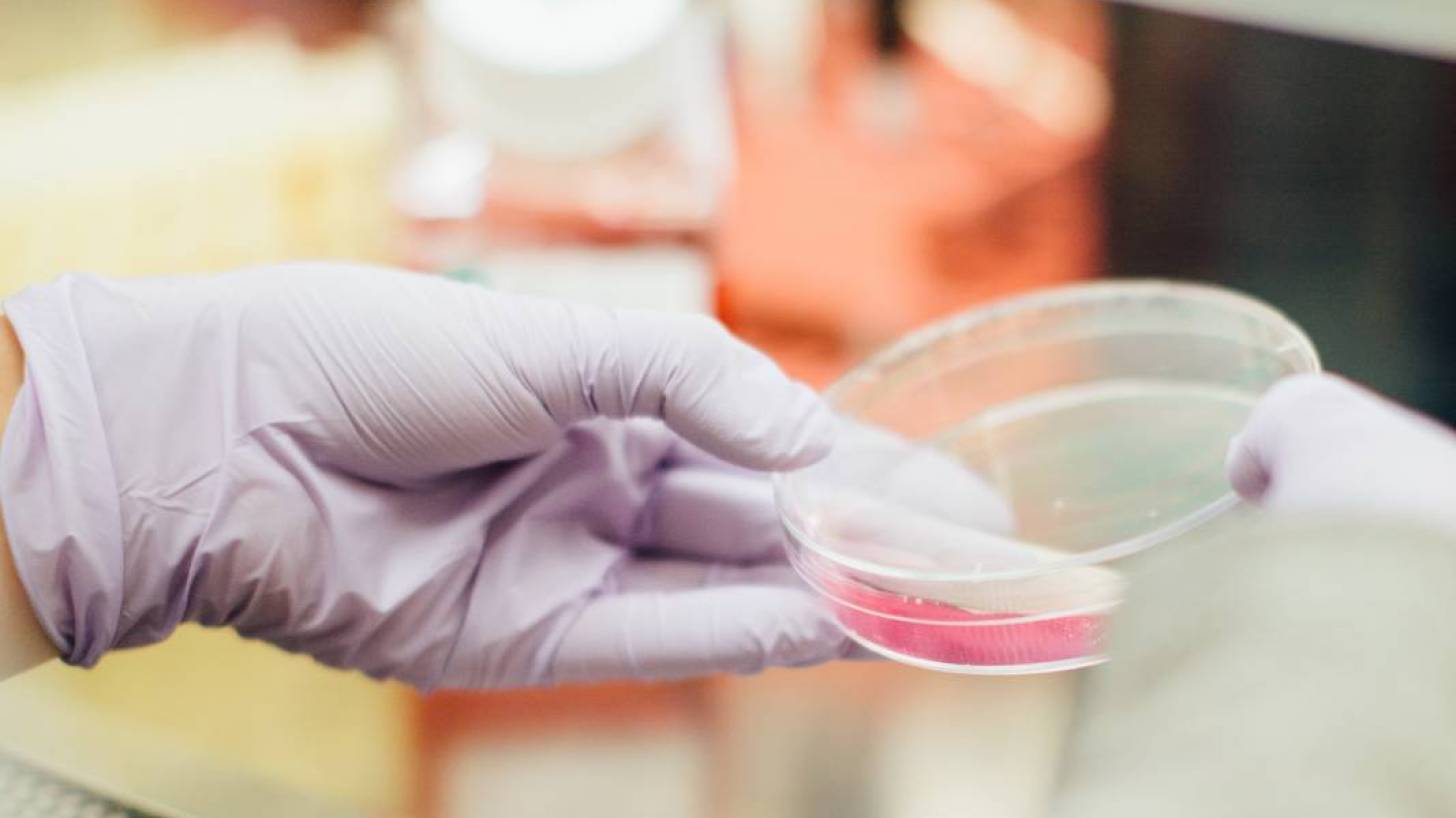
Colorado researchers announced they have developed a rapid, portable, saliva-based COVID-19 disease test able to return results in 45 minutes.
Because no swabs or fancy equipment is needed, the new tests are less vulnerable to backlogs and supply chain shortages, the researchers say.
The test is based on a 20-year-old technology known as reverse transcription loop-mediated isothermal amplification (RT-LAMP) previously used in remote regions of South America.
“We are facing a serious testing shortage in this country right now as more people want to get tested and diagnostics labs are overwhelmed,” said Nicholas Meyerson, a postdoctoral associate in the Sawyer Lab at the BioFrontiers Institute at Colorada University (CU) at Boulder, in a press statement issued on July 22, 2020.
The COVID-19 diagnostic test, described in a preprint manuscript of the non-peer-reviewed study by MedRxiv.org on July 17, 2020, is designed for widespread screening to help identify asymptomatic individuals.
Previous research shows people infected with the betacoronavirus, but with no obvious symptoms, makeup as many as 70 percent of cases and can still spread disease.
In this new CU test, a user spits in a tube, adds a solution to stabilize it then closes the lid and hands it off to testing staff. They process it through a simple system requiring little more than pipettes, a heating source, and an enzyme mixture.
If the sample turns from pink to yellow, the test is positive. If it doesn’t, it’s negative.
“Every test that has been approved to date requires that the sample, even if it’s saliva, be processed in a clinical diagnostic lab or at a doctor’s office, using sophisticated equipment. That can take up to nine days right now,” said CU Professor Sara Sawyer, a virologist in the Department of Molecular Cellular and Developmental Biology, who led the development of the test.
Once a sample is collected, it is heated to liberate any viral genome present in the test liquid. This sample is then added to three tubes, each containing a custom enzyme mixture which, when heated to a certain temperature, undergoes a chemical reaction when the genetic material from SARS-CoV-2 is detected.
That’s the virus that causes COVID-19 disease.
In one experiment described in the paper, the researchers conducted what is known as a “contrived clinical validation.”
One researcher spiked 30 out of 60 saliva samples with inactivated SARS-CoV-2 in the lab. Then they shuffled the samples and gave them to another scientist to test with the RT-LAMP technology.
“The test predicted with 100% accuracy all of the negative samples, and 29 of 30 positive samples were predicted accurately,” said Meyerson, noting that the 30th test was scored as inconclusive. Additional second-party validation tests are currently underway.
The authors note that the test is slightly less sensitive than those performed in clinical labs. But a separate computer modeling study, also by researchers at the BioFrontiers Institute, found that quick turnaround for testing is even more critical to curbing the pandemic than test sensitivity is.
“Our modeling showed that whether a test is sensitive or super-sensitive is not that important,” said BioFrontiers Director Roy Parker, co-author of that paper.
“What is important is frequent testing, with the test results returned as fast as possible, which identifies more infected people faster and can limit new infections.”
“Those who test positive could quarantine themselves as they await confirmatory testing,” said Sawyer.
The CU team is also working with the state to carry out further validation tests locally and, pending those results, expedite regulatory clearance for use in Colorado.
The research team, in cooperation with Venture Partners at CU Boulder, has created a spinoff company, Darwin Biosciences, to commercialize the test. The test has not been approved by the Food and Drug Administration yet, but the team has submitted paperwork via the agency's Emergency Use Authorization program.
"We are very excited and applaud the scientists at the University of Colorado," said Governor Jared Polis, during a press briefing on July 23rd.
Study funding: the Burroughs Wellcome Fund and NIH. Furthermore, no research conflict of interest was disclosed.
Precision Vaccinations publishes diagnostic test news.
Our Trust Standards: Medical Advisory Committee

























