Marburg Virus Vaccine Candidate Launches Phase 1 Study In Maryland

A phase 1 clinical trial evaluating a Marburg vaccine candidate administered its first vaccine dose in early October 2018.
The VRC-MARADC087-00-VP vaccine candidate being tested was developed by the Vaccine Research Center (VRC) of the National Institute of Allergy and Infectious Diseases (NIAID).
This recombinant chimpanzee adenovirus type 3-vectored Marburg virus vaccine candidate is aimed at stimulating rapid, but durable immunity.
This vaccination strategy is considered optimal to protect both first-line health workers and general populations when outbreaks occur.
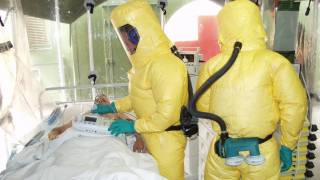
"Given the recent deadly outbreak of Marburg virus in Uganda in 2017, there is a critical need to develop a safe and effective vaccine which has the potential to protect our soldiers and the public from this serious threat," said Lt. Col. Melinda Hamer, chief of the Walter Reed Army Institute of Research (WRAIR) Clinical Trials Center, in a press release.
This WRAIR study is being conducted in Silver Spring, Maryland.
Only 40 healthy, adult volunteers will be enrolled and assigned to one of two groups to evaluate different vaccine dosages. Volunteers will be closely followed over the course of approximately one year after vaccination for safety and immune system response.
Marburg virus was first recognized in 1967, when outbreaks of hemorrhagic fever occurred simultaneously in laboratories in Marburg and Frankfurt, Germany and in Belgrade, Serbia.
The reservoir host of Marburg virus is the African fruit bat, Rousettus aegyptiacus.
Case fatality rates in Marburg outbreaks have ranged from 24 to 88 percent, says the Centers for Disease Control and Prevention.
The Marburg virus is in the same family as Ebola and causes severe hemorrhagic fever in humans. WRAIR conducted the first Ebola vaccine clinical trial in Africa in 2009, testing an early vaccine candidate also developed by the VRC at NIAID.
During the 2014 West Africa Ebola outbreak, WRAIR conceived of and then initiated a Phase I clinical trial of the VSV-EBOV vaccine candidate over a period of just 11 weeks. WRAIR has conducted six Ebola vaccine studies to date.
"This first-in-human trial of the NIH vaccine is part of an important inter-agency effort by the US government to anticipate and counter emerging infectious threats to the Americans, both military and civilian, at home and abroad," added Dr. Kayvon Modjarrad, the director of the Emerging Infectious Diseases Branch at WRAIR.
Our Trust Standards: Medical Advisory Committee