Zaire Ebolavirus Vaccine Approved for More Children

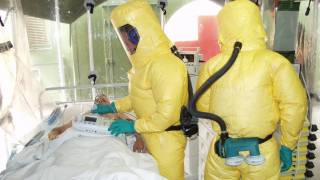
Since Ebola virus disease (EVD) impacts everyone in an outbreak area, another agency has approved a preventive vaccine for young children.
Merck today announced that the European Commission (EC) approved an expanded indication for the ERVEBO® vaccine for active immunization of individuals one year of age or older to protect against EVD caused by Zaire ebolavirus.
ERVENO was previously approved for use in the European Union for individuals 18 or older.
The U.S. FDA recently issued similar approval for young children.
ERVEBO is approved in the European Union, United Kingdom, United States, Canada, Switzerland, and ten countries in Africa.
ERVEBO is a live recombinant viral vaccine with a vesicular stomatitis virus backbone that protects people from Zaire ebolavirus. This vaccine does not protect people from the Sudan ebolavirus or the Marburg virus.
Dr. Eliav Barr, senior vice president, head of global clinical development, and chief medical officer, Merck Research Laboratories, commented in a press release on September 7, 2023, “When outbreaks of Ebola virus disease occur, they can quickly become a public health crisis. We are proud to play a role, alongside the global public health community, in helping to prepare for potential outbreaks of Zaire ebolavirus.”
In January 2021, Merck confirmed an agreement with UNICEF to establish the world’s first global Ebola vaccine stockpile to support future Zaire ebolavirus (EBOV) outbreak preparedness and response efforts.
Over 500,000 doses of ERVEBO have been delivered to the stockpile, administered by the International Coordinating Group on Vaccine Provision.
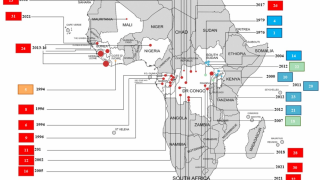
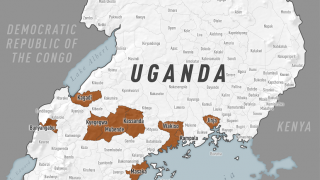
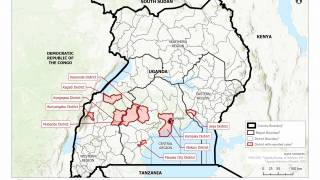
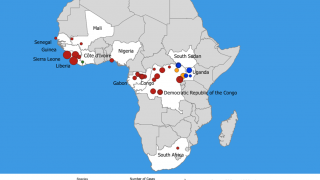
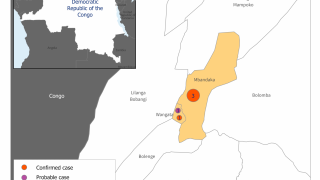
The initial EBOV case was confirmed in 1976 in the African countries of South Sudan and the Democratic Republic of Congo. Recent data suggest EBOV outbreaks may originate from human-to-human transmission instead of spillover events.
The U.S. Centers for Disease Control and Prevention published a list of EBOV Cases and Outbreaks as of August 2023.
Our Trust Standards: Medical Advisory Committee