Yellow Fever Vaccines
Yellow Fever Vaccines 2024
Effective yellow fever vaccines are available in Europe, the United States, and the United Kingdom in 2024 and have been Listed by the World Health Organization (WHO) for decades. According to the U.S. Centers for Disease Control and Prevention (CDC), yellow fever vaccination is recommended for people aged nine months or older who are traveling to or living in areas at risk for the yellow fever virus. The 17D yellow fever vaccine was initially approved for human use in 1938, with about 850 million doses distributed worldwide, reported a study in Nature. According to the WHO, the yellow fever vaccine coverage in countries at risk of the disease is about 48%. In 2023, nearly 62 million individuals received vaccinations under the EYE strategy.
The WHO, the European Medicines Agency (EMA), the Pan American Health Organization (PAHO), and the CDC say a single yellow fever vaccination can grant life-long protection and promote sustained immunity for about 90% of people vaccinated.
Yellow Fever Vaccine Availablity April 2024
These manufacturers produce about 80 million doses annually. Reports in 2023 indicate a six million 17D-213 vaccine dose capacity in the global stockpile.
YF-VAX® vaccine is licensed in the U.S. (BL 103915) and available at certified travel vaccine clinics and pharmacies.
Stamaril® vaccine is available internationally in about 40 countries.
Bio-Manguinhos Yellow fever vaccine - The National Immunization Program offers the yellow fever vaccine in Brazil.
Institut Pasteur Yellow fever vaccine is produced at the Institut Pasteur in Dakar. The WHO approved the laboratory to supply the yellow fever vaccine to Expanded Programs on Immunization in Africa.
Chumakov Institute Yellow fever vaccine - The Chumakov Center is a research organization in medical virology. The vaccine is a freeze-dried virus-containing suspension of specific pathogen-free chick embryo tissues infected with attenuated Yellow Fever virus, strain 17D.
RegaVax is a vaccine candidate using the yellow fever vaccine virus's genetic code as a carrier (or vector) for the coronavirus's genetic code.
Najít Technologies HydroVax-002 YFV is a Novel Inactivated Yellow Fever vaccine candidate conducting phase 1 clinical research with the NIH. The study evaluated 1 mcg and 5 mcg dosages were given intramuscularly on Day 1 and Day 29.
SII YFV containing the 17D-213 vaccine strain has been developed in India. A Phase I study was conducted to evaluate the safety and immunogenicity of healthy adult volunteers. This YFV was found safe and immunogenic by IM and SC routes.
Yellow Fever Vaccine Boosters
The Lancet Global Health published results from a systematic review and meta-analysis in January 2024 focused on determining the long-term immunity following yellow fever vaccination. The evidence suggests that a single dose of yellow fever vaccination provides lifelong protection for travelers. However, in people living with HIV and children (younger than two years), booster doses might still be required because lower proportions of vaccinees were seroprotected ten or more years post-vaccination. Pooled seroprotection rates were 94% (95% CI 86–99) among healthy adults in a non-endemic setting (mostly travelers) and 76% (65–85) in an endemic setting (all Brazilian studies). The pooled seroprotection rate was 47% (35–60) in children (aged 9–23 months at the time of vaccination) and 61% (38–82) in people living with HIV.
The U.S. CDC says a yellow fever vaccine booster dose is unnecessary.
Yellow Fever Vaccine Fractional Dosage
The peer-reviewed journal The Lancet Infectious Diseases published an Opinion on April 28, 2023, saying fractional dose yellow vaccination in an emergency or reactive mass vaccination campaign has become essential in stretching the limited vaccine stockpile over a much larger population. On April 28, 2023, The Lancet published phase 4 clinical study results from immunogenicity and safety of fractional doses of 17D-213 yellow fever vaccine in HIV-infected people in Kenya, that concluded fractional doses of the 17D-213 yellow fever vaccine were sufficiently immunogenic and safe, demonstrating non-inferiority to the standard vaccine dose in HIV-infected individuals with CD4+ T cell counts of at least 200 cells per mL. These results confirm that fractional dose recommendations apply to populations with high HIV prevalence.
The Lancet published results from a clinical study in January 2021 (updated Feb. 2022, concluding, 'Fractional doses of all WHO-prequalified yellow fever vaccines were non-inferior to the standard dose in inducing seroconversion 28 days after vaccination, with no major safety concerns. These results support using fractional dosage in the general adult population for outbreak response in a vaccine shortage.' The WHO's initial position on fractional yellow fever vaccine was set out in the 2013 WHO position paper.
Yellow Fever Vaccination Card
The International Certificate of Vaccination or Prophylaxis (ICVP), known as the yellow card, is required to enter certain countries. For example, in the U.S., vaccinated people receive a yellow fever card (ICVP) to prove they recently had yellow fever vaccinations, says the WHO. For specific information on ICVP entry requirements, please see the individual country pages, such as Argentina, Angola, Bolivia, Brazil, Ecuador, Ghana, Kenya, Paraguay, Rwanda, and Uganda. In addition, ICVP requirements are contained in the WHO's Country List. The U.K.'s National Health Service (NHS) updated yellow fever vaccination requirements for specific countries in November 2022, and requirements are available on the CDC Travelers' Health webpage.
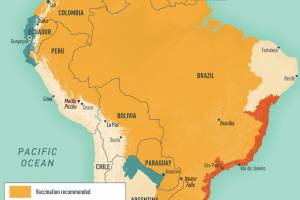
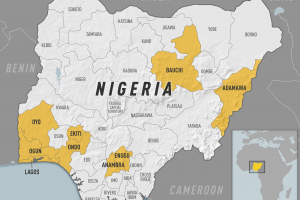
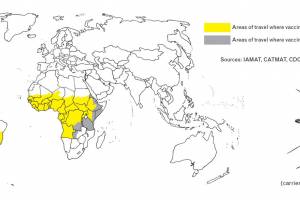
African countries that require proof of YF vaccination from all arriving travelers: Angola, Benin, Burkina Faso, Burundi, Cameroon, Central African Republic, Congo, Republic of the Côte d'Ivoire, Democratic Republic of the Congo, Gabon, Ghana, Guinea, Guinea-Bissau, Mali, Niger, Sierra Leone, South Sudan, Togo, Argentina, Bolivia, Brazil, Colombia, Ecuador, French Guiana, Guyana, Panama, Paraguay, Peru, Suriname, Trinidad and Tobago, and Venez are the countries in the American region requiring proof of yellow fever vaccination.
Eliminate Yellow Fever Epidemics Strategy
The Eliminate Yellow Fever Epidemics (EYE), a global coalition of over 50 partners in 40 countries, seeks to vaccinate nearly one billion people against yellow fever by 2026. Since 2017, according to EYE Strategy data in 2022, 226 million more people in Africa have been protected from yellow fever via a single-dose vaccine.
Yellow Fever Vaccine Price
The CDC's Vaccines For Children program provides vaccines to children at no cost. Additionally, UNICEF publishes vaccine prices for children. As is the Stamaril vaccine price, outside of the U.S., Sanofi's Patient Connection® offers various vaccine price savings. In addition, Sanofi has live support specialists at (800) 633-1610 to answer patients' questions. And InstantRx™ is a digital gateway empowering people to find lower-priced vaccines easily.
Yellow Fever Travel Advisories
The U.S. CDC and the U.K. Travel Health Advice recently issued yellow fever Travel Advisories. As of April 2024, yellow fever outbreak news is posted at Precision Vax.