Chikungunya Vaccine VLA1553 ‘Fast-Tracked’ By FDA

The U.S. Food and Drug Administration (FDA) has granted Fast Track designation for the chikungunya vaccine candidate, VLA1553.
VLA1553 is a monovalent, single-dose, live-attenuated vaccine candidate for protection against chikungunya. It is designed for prophylactic, active, immunization in humans over 1 year of age.
In pre-clinical development, a single-dose of VLA1553 was reported to be highly immunogenic in vaccinated Non-Human Primates and showed no signs of viremia after challenge.
Fast Track designation facilitates the clinical development and expedites the review of new drugs and vaccines with the intention of accelerating the availability of promising products on the market, says the FDA.
There are no preventive vaccines or effective treatments available and, as such, chikungunya is considered to be a major public health threat.
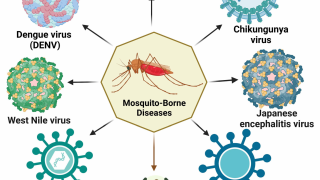
Chikungunya is a mosquito-borne viral disease caused by the chikungunya virus (CHIKV), a Togaviridae virus, transmitted by Aedes mosquitoes, says the Centers for Disease Control and Prevention (CDC).
Clinical symptoms include acute onset of fever, debilitating joint and muscle pain, headache, nausea, and rash, potentially developing into long-term, serious health impairments.
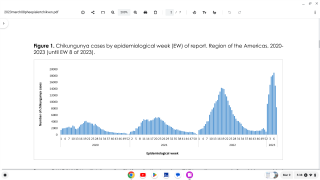
Chikungunya virus causes clinical illness in 72% to 92% of infected humans around four to seven days after an infected mosquito bite.
Fatalities have been reported (case fatality rates of 0.1% to 4.9% from epidemics) in elderly patients, who are at a higher risk.
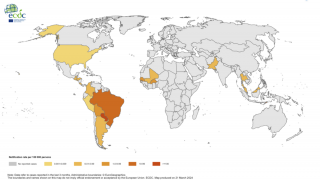
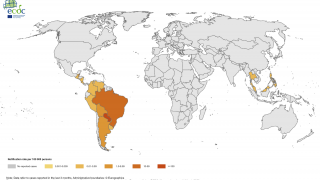
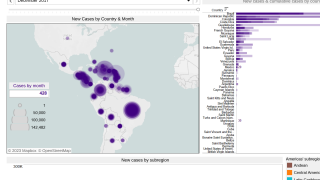
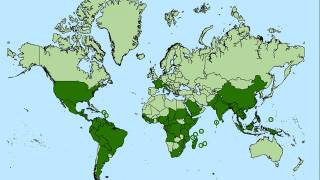
Outbreaks have occurred in Africa, Asia, Europe, and the Indian and Pacific Oceans. During 2013, chikungunya virus was found for the first time in the Americas on islands in the Caribbean.
Valneva SE said in a press release it will release VLA1553's initial Phase 1 data in the coming weeks.
Wolfgang Bender, MD, Ph.D., Chief Medical Officer of Valneva SE, commented in a press release, "The fast track designation will allow us to work closely with the FDA and to accelerate our efforts to develop a one dose solution for the prevention of this spreading disease."
Valneva SE is a biotech company developing and commercializing vaccines for infectious diseases with major unmet needs. More information is available at Valneva.com.
Our Trust Standards: Medical Advisory Committee