TB Vaccine Candidate Offered Potential for BCG Revaccination Strategies
A nonprofit organization developing vaccines against tuberculosis (TB) announced a prevention-of-infection Phase 2 clinical trial revaccination with the Bacille Calmette-Guerin (BCG) vaccine, significantly reduced sustained TB infections in adolescents.
Introduced in 1921, BCG is the only vaccine currently licensed to prevent TB disease.
An experimental vaccine candidate, H4:IC31, is the first time a subunit vaccine has shown any indication of ability to protect against TB infection or disease in humans.
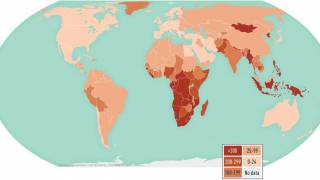
According to the World Health Organization (WHO), about 30 percent of the world's population has what is called a latent TB infection.
This means people have been infected by TB bacteria, but have not been impacted by the disease.
People infected with TB bacteria have a lifetime risk of falling ill with TB of 10 percent.
People ill with TB can infect 10-15 other people through close contact over the course of a year.
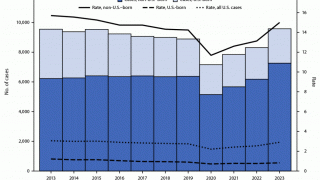
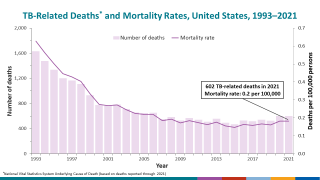
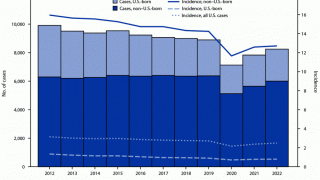
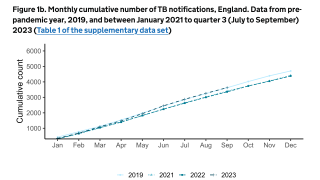
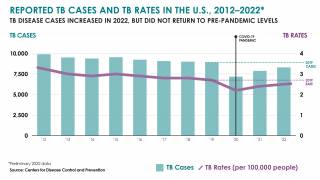
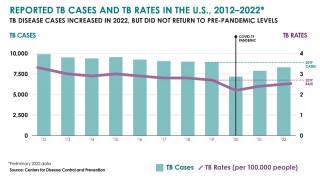
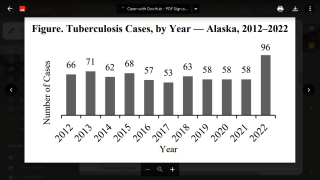
Tuberculosis causes more deaths than any other single infectious disease and is increasingly characterized by antimicrobial resistance. There were 10.4 million new cases of TB in 2016 and 1.7 million deaths, according to the WHO.
Mark Hatherill, MD, Director of the South African Tuberculosis Vaccine Initiative (SATVI) at the University of Cape Town, and the study's principal investigator, said: "We are pleased to have performed the first-known randomized, placebo-controlled prevention-of-infection trial for TB and to have demonstrated that vaccination has the potential to reduce the rate of sustained TB infection in a high-transmission setting.’
“We are extremely encouraged by the signals observed for both vaccines in preventing sustained TB infections.”
The study evaluated H4:IC31 vaccination and BCG revaccination for safety, immunogenicity and the ability to prevent initial and sustained TB infections among healthy adolescents in the Western Cape Province of South Africa.
For the primary efficacy endpoint, 134 participants tested positive for an initial Mtb infection as measured by QFT-GIT conversion from negative to positive (placebo=49; BCG=41; H4: IC31=44). When compared to placebo, neither vaccine achieved statistical significance in preventing an initial QFT-GIT conversion. Observed vaccine efficacy was 20.1% (p=0.15) for BCG revaccination and 9.4% (p=0.32) for H4:IC31.
For the secondary efficacy endpoint, 82 participants exhibited a sustained QFT-GIT conversion lasting at least 6 months following initial conversion (placebo=36; BCG=21; H4:IC31=25). These participants were evaluated to see if they would revert to negative as measured by the QFT-GIT test. In the BCG revaccination arm, the vaccine efficacy for preventing a sustained infection was 45.4% (p=0.013) and in the H4:IC31 arm the vaccine efficacy was 30.5% (p=0.08).
The clinical trial was conducted at SATVI and at the Emavundleni Research Centre (part of the Desmond Tutu HIV Centre). It was funded by Sanofi Pasteur, the United Kingdom's Department for International Development, and Aeras. The study was approved by the Medicines Control Council of South Africa and the relevant local independent ethics committees.
The data showed that both vaccines appeared to be safe and produced an immune response in the adolescents studied.
No vaccine-related serious adverse events were reported in the study, and the most common vaccine-related adverse event was injection site swelling in BCG revaccinated participants, typical for BCG vaccination.
Our Trust Standards: Medical Advisory Committee