Hepatitis A Health Advisory Issued by CDC

The Centers for Disease Control and Prevention (CDC) issued an official Health Advisory on June 11, 2018, regarding the ongoing hepatitis A outbreaks.
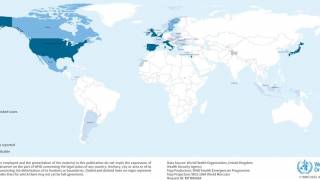
From January 2017 to April 2018, the CDC has received more than 2,500 reports of hepatitis A infections associated with person-to-person transmission.
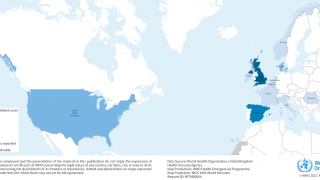
These hepatitis A cases have been reported in various states, primarily clustered in the Midwest.
Of the more than 1,900 reports for which risk factors are known, more than 1,300 (68%) of the infected persons report drug use, homelessness, or both.
This data compares with the mid-1980s when drug use was a risk factor for less than 20 percent of all hepatitis A cases reported to CDC.
Outbreaks of hepatitis A infections among homeless persons have occurred in other countries, but large outbreaks among the homeless have not been described previously in the United States.
Hepatitis A outbreaks among these groups are difficult to control.
An HAV-infected person can be viremic up to 6 weeks through their clinical course and excrete virus in stool for up to 2 weeks prior to becoming symptomatic, making identifying exposures particularly difficult.
The state and local health departments in affected areas are recommending vaccination of individuals at highest risk of acquiring the infection.
But, the CDC does not recommend vaccinating all food handlers because doing so would not prevent or stop the ongoing outbreaks primarily affecting individuals who report using illicit drugs and people experiencing homelessness.
The best way to prevent hepatitis A infection is through vaccination with the hepatitis A vaccine. The number and timing of the doses depend on the type of vaccine administered.
Inactivated vaccines containing HAV antigen are currently licensed and available in the United States are:
- Single-antigen vaccines HAVRIX®(manufactured by GlaxoSmithKline, Rixensart, Belgium) and VAQTA® (manufactured by Merck & Co., Inc., Whitehouse Station, New Jersey) and,
- Combination vaccine TWINRIX® (containing both HAV and hepatitis B virus antigens; manufactured by GlaxoSmithKline).
- GamaSTAN S/D (Grifols Therapeutics, Inc., Research Triangle Park, North Carolina) immune globulin (IG) for intramuscular administration is the only IG product approved for HAV prophylaxis.
Most pharmacies in the USA offer these hepatitis vaccines.
To schedule a vaccination appointment at a local pharmacy, please click here.
The CDC Vaccine Price List provides current HAV vaccine contract prices and general information, and vaccine discounts can be found here.
Vaccines, like any medicine, can have side effects, says the CDC. You are encouraged to report negative side effects of vaccines to the FDA or CDC.
Our Trust Standards: Medical Advisory Committee