Tuberculosis Vaccines Do Work, Occasionally

Aeras, a nonprofit organization, announced the publication of the full results from a Phase 2, randomized, controlled clinical trial of 2 tuberculosis (TB) vaccines.
This proof-of-concept study showed that vaccination can reduce the rate of sustained TB infections in a high-transmission setting, but lacked full efficacy against initial Mycobacterium tuberculosis infection.
In this trial, revaccination with BCG significantly reduced sustained TB infections in adolescents with a 45.4 percent vaccine efficacy.
Additionally, the investigational vaccine, H4: IC31, also reduced sustained infections, although not at statistically significant levels, showing 30.5% vaccine efficacy.
This means neither vaccine met the primary endpoint of initial prevention.
However, the trend observed for H4: IC31 is the first time a subunit vaccine has shown any signal that it may be able to protect against TB infection or disease in humans.
Because there is no definitive test for M. tuberculosis infection, these researchers said the clinical implications of the findings are limited.
Jacqueline Shea, Ph.D., Chief Executive Officer at Aeras, said: “With this study, we showed that vaccines against TB infection can work.”
“The results highlight the importance of investing in new approaches to fighting the leading infectious disease killer and to evaluating new concepts in clinical trials.”
“The BCG results are important findings with significant public health implications that could lead to saving millions of lives,” said Dr, Shea.
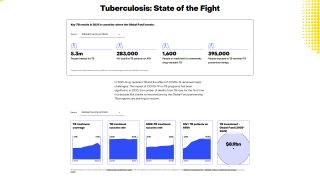
Tuberculosis causes more deaths than any other single infectious agent and is increasingly characterized by antimicrobial resistance. There were 10.4 million new cases of TB in 2016 and 1.7 million deaths, reports the WHO.
BCG is the only licensed tuberculosis vaccine available globally.
H4: IC31 is an investigative subunit vaccine candidate being developed jointly by Aeras and Sanofi Pasteur, and the Statens Serum Institut.
The clinical trial was funded by Sanofi Pasteur, the United Kingdom’s Department for International Development, The Bill & Melinda Gates Foundation and Aeras.
The clinical trial was conducted at the South African Tuberculosis Vaccine Initiative (SATVI) at the University of Cape Town and at the Emavundleni Research Centre (part of the Desmond Tutu HIV Centre). The study was approved by the Medicines Control Council of South Africa and the relevant local independent ethics committees.
Our Trust Standards: Medical Advisory Committee