Remdesivir to Treat Ebola Virus Infections

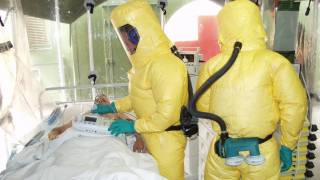
There is some good and bad news regarding the ongoing Ebola Zaire virus outbreak in the Democratic Republic of Congo.
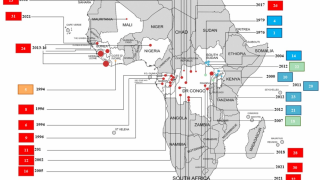
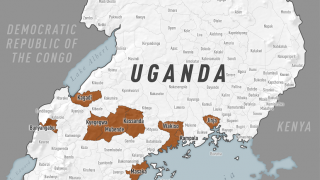
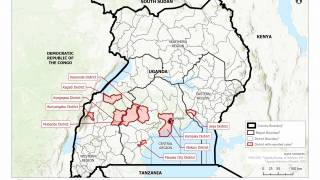
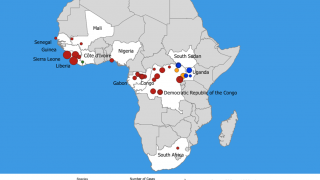
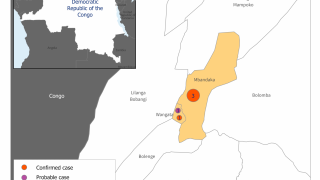
The bad news is this Ebola outbreak has now claimed 660 lives since beginning in August 2018.
The good news announced on March 29, 2019, by USAMRIID, for the first time, the U.S. Food and Drug Administration (FDA) has provided a formal regulatory agreement for use of an animal model to support the development of the drug candidate, Remdesivir.
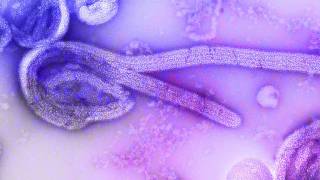
Remdesivir is an investigational agent for treating deadly Ebola virus (EBOV) infections and is not approved by any regulatory agency globally.
Its safety and efficacy have not been established, says Gilead Sciences, Inc.
In 2014, Gilead began a collaboration with the Centers for Disease Control and Prevention and the U.S. Army Medical Research Institute for Infectious Diseases (USAMRIID) to see whether several molecules originally explored as potential treatments for other viruses including hepatitis C, respiratory syncytial virus, and influenza might also be active against emerging viruses such as Ebola.
The collaboration identified as Remdesivir (GS-5734), is a nucleotide prodrug that is processed in the body to rapidly release the active drug into cells.
Specifically, the FDA agreed that the rhesus macaque, infected by intramuscular (IM) injection, is a relevant and adequately characterized model of Ebola virus disease to support filing under the FDA Animal Rule.
In addition, the FDA agreed that the rhesus IM/EBOV disease model is sufficient as a single animal model for therapeutic product development.
Notably, the FDA also agreed that a specific delayed time-to-treat approach is appropriate for future nonclinical studies aimed at characterizing the efficacy of Remdesivir.
The U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID) and Gilead Sciences, Inc. worked in close partnership to develop the study plan for conducting the IM/EBOV NHS in rhesus monkeys, analyze the study outcome and submit data to the FDA.
"To date, there are no FDA-approved therapeutics for the treatment of Ebola virus disease. Both the current outbreak in the Democratic Republic of Congo and the 2014-2016 West Africa outbreak, the largest in history, highlight the urgent need for antiviral therapy to combat this deadly disease,” said John McHutchison, AO, MD, Chief Scientific Officer and Head of Research and Development, Gilead Sciences, Inc.
Recent Ebola news:
- Ebola Outbreak in Africa Reports 629 Deaths
- Ebola Vaccine Candidate Reported 96% Seroconverted after 3 Doses
- CDC Expands Level 2 Travel Alert for the Democratic Republic of Congo
Our Trust Standards: Medical Advisory Committee