Nasal Whooping-Cough Vaccine Launches Clinical Trial

Researchers at Vanderbilt University Medical Center have begun enrolling volunteers in a Phase 2 clinical trial that will study a next-generation pertussis vaccine that may protect people from ‘whooping cough’.
The BPZE1 vaccine candidate is a live, attenuated form of the pertussis bacterium.
This innovative vaccine is given as a mist into the nose, rather than a shot in the arm.
Buddy Creech, MD, MPH, director of the Vanderbilt Vaccine Research Program, said in a press release, “Administering the vaccine into the nose, instead of giving a traditional shot, may more closely mimic what is seen in a natural infection.”
“It only makes sense that, when we can, we try to deliver vaccines to the place in the body where we first see the germ.”
“For pertussis, that is the nose,” Dr. Creech said.
“What we are learning is that the vaccine now in use is good at preventing disease, but other vaccines may have a greater duration of protection and be better in preventing the germ from being carried in the nose altogether or being transmitted from one person to another,” he said.
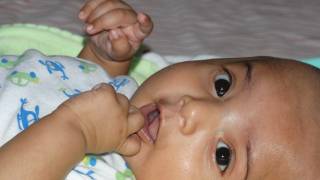
Pertussis is known for uncontrollable, violent coughing which often makes it hard to breathe. After cough fits, someone with pertussis often needs to take deep breaths, which result in a “whooping” sound.
Pertussis can affect people of all ages, but can be very serious, even deadly, for babies less than a year old, says the Centers for Disease Control and Prevention (CDC).
Recent pertussis vaccine news:
- 1st Tdap Vaccine Approved for a 2nd Dose
- Tdap Vaccine Delivers Best Protection From Whooping Cough Between 27 and 30 Weeks
- Pregnant Women in California Need Better Tdap Vaccination Management
ILiAD Biotechnologies, LLC, (ILiAD) is providing the vaccine for this clinical study. ILiAD is a clinical-stage biotech company that announced in December 2018 it had secured up to $20 million of additional investment.
During May 2018, the National Institute for Allergy and Infectious Diseases (NIAID) and ILiAD signed a clinical trial agreement calling for NIAID to sponsor the BPZE1 Phase 2a clinical trial and for ILiAD to supply BPZE1 vaccine study product.
Our Trust Standards: Medical Advisory Committee