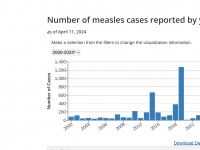
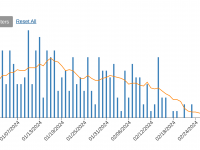
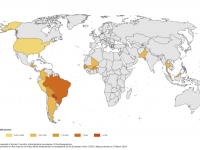
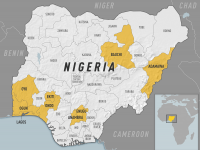
ImmunityBio, Inc. today announced that the U.S. Food and Drug Administration (FDA) had approved ANKTIVA® (N-803) plus Bacillus Calmette-Guérin (BCG) vaccine for the treatment of patients with BCG-unresponsive non-muscle invasive bladder cancer (NMIBC) with carcinoma in... READ →
Precision Vaccinations
/ April 23rd, 2024Eliminating fake vaccine news with pharmacist, nurse, and physician review.
For more articles, try our search.