Ebola Vaccine Candidates Generating Positive Immune Responses

Ever since the development of the first vaccine more than 200 years ago, vaccinations have greatly decreased the burden of infectious diseases, such as the Ebola virus.
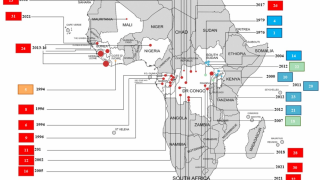
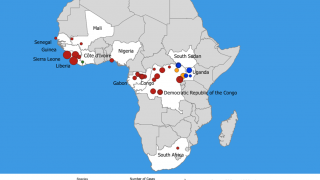
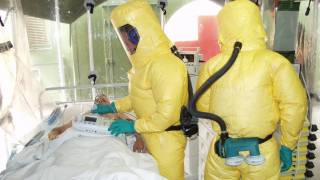
The Ebola virus disease has become a great threat to humans in recent years.
New research has reported that the 3 Ebola virus vaccine candidates generate an immune response against the virus that lasts for at least 2.5 years.
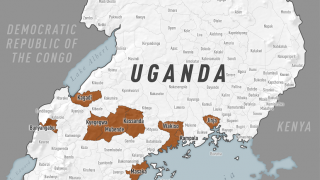
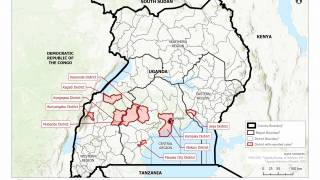
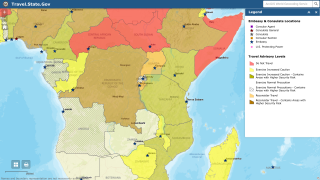
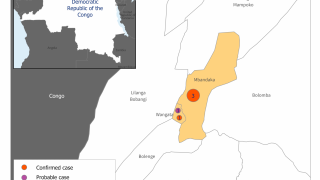
This is good news given the ongoing Ebola outbreak in the Democratic Republic of Congo, which has reported 139 fatalities since August 1, 2018.
The study, which is being presented at the Annual Meeting of the American Society of Tropical Medicine and Hygiene, ‘could have implications far beyond the Ebola fight,’ said Katie Ewer, Ph.D., who conducted the study, in a press release.
Dr. Ewer said, “The impact could help expedite vaccines against several other diseases with significant outbreak potential, including Lassa fever, Nipah virus disease, and the Middle East Respiratory syndrome coronavirus (MERS-CoV).”
"Another important question is whether the persistence of this immune response can be enhanced by giving a 'late-booster' dose of vaccine 3 to 4 years after the initial immunization,” said Dr. Ewer.
Dr. Ewer said “In general, the analysis of the experimental Ebola vaccines showed that all three "were still producing a strong antibody response to the disease two and a half years after immunization, which is really good news."
One of the vaccines, developed by Merck, is now being used in the DRC, and there was already preliminary evidence that its protection lasted for at least 2 years.
While the protection from the vaccines might actually be much longer, this is the longest scientists have been able to track Ebola vaccine responses in humans.
Ewer said that "to protect health-care workers in the field in regions where we know there is a risk of new Ebola outbreaks, it's really important to generate persistent immune responses and this is now the most urgent need for an Ebola vaccine."
The goal now said Dr. Ewer, “is to have more vaccines that can be quickly readied for at least limited use at the first sign of an outbreak--to quickly protect healthcare workers and contain the spread with a ring of immunized individuals, as well as protecting against future outbreaks.”
Recently, the WHO says there is not enough valid research to risk the health of pregnant women nor their future child with the investigational vaccine rVSV-ZEBOV.
The study was funded by an Innovate U.K. award from the U.K. Department of Health to Matthew Snape.
The American Society of Tropical Medicine and Hygiene, founded in 1903, is the largest international scientific organization of experts dedicated to reducing the worldwide burden of tropical infectious diseases and improving global health.
Media Contacts: Preeti Singh [email protected] and Bridget DeSimone [email protected]
Our Trust Standards: Medical Advisory Committee