15-Valent Pneumococcal Conjugate Vaccine Granted Breakthrough Status

Merck’s investigational 15-valent pneumococcal conjugate vaccine V114 has received Breakthrough Therapy Designation from the U.S. Food and Drug Administration (FDA) for the prevention of invasive pneumococcal disease (IPD) caused by the vaccine serotypes in pediatric patients 6 weeks to 18 years of age.
To qualify for this FDA designation, preliminary clinical evidence must demonstrate that the drug may provide a substantial improvement over currently available therapy on at least one clinically significant endpoint.
V114 is also under development for the prevention of IPD in adults. Both indications are currently being studied in Phase 3 clinical trials.
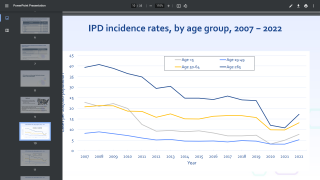
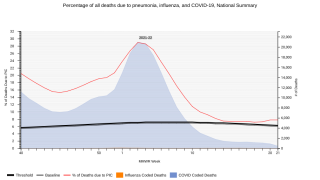
This is important news since the World Health Organization estimates that S. pneumoniae kills close to 500,000 children under 5 years old worldwide every year, with most of these deaths occurring in developing countries.
The FDA’s decision was informed in part by immunogenicity data from 2 studies, which are as follows:
- Study 005 was a Phase 1/2, multicentre, randomized, double-blind study to evaluate the safety, tolerability and immunogenicity profiles of 4 different lots of a new formulation of V114 in healthy adults and infants.
- Study 008 was a proof of concept, Phase 2, pediatric trial to confirm the results from Study 005 in a larger population of infants.
In both studies, V114 induced an immune response in infants for 2 disease-causing serotypes (22F and 33F) not contained in the currently available 13-valent pneumococcal conjugate vaccine, while demonstrating non-inferiority for the serotypes contained in both vaccines.
Invasive pneumococcal disease is a notable cause of morbidity and mortality in the US, despite the availability of 7-valent pneumococcal conjugate vaccine (PCV7) and 13-valent pneumococcal conjugate vaccine (PCV13), says the Centers for Disease Control and Prevention (CDC).
Dr. Nicholas Kartsonis, senior vice president and head of vaccine and infectious diseases clinical research at Merck Research Laboratories, said in a press release, “The goal of our program in pediatric patients remains focused on providing additional serotype coverage versus currently available vaccines, while at the same time maintaining a strong immune response across all serotypes in the vaccine.”
Streptococcus pneumoniae, or pneumococcus, is a type of bacterium that causes pneumococcal disease. Pneumococcal infections can range from ear and sinus infections to pneumonia and bloodstream infections.
Children younger than 2 years old and adults 65 years or older are among those most at risk for the disease. There are vaccines to prevent pneumococcal disease in children and adults, says the CDC.
Merck is a leading global biopharmaceutical company known as MSD outside of the United States and Canada. For more information, visit Merck.
Our Trust Standards: Medical Advisory Committee
- Merck Receives Breakthrough Therapy Designation from FDA for V114, the Company’s Investigational 15-Valent Conjugate Vaccine
- A Phase I-II, Randomized, Double-Blind, Study to Evaluate the Safety, Tolerability, and Immunogenicity of Different Formulations
- Safety and immunogenicity of 15-valent pneumococcal conjugate vaccine compared to 13-valent pneumococcal conjugate vaccine
- Global Pneumococcal Disease and Vaccine